|
Stephen Hauschka
Emeritus Professor of Biochemistry
Adjunct Professor of Zoology
BA, Amherst College
PhD, Johns Hopkins University
|
Research
Research in our group addresses basic questions of skeletal and cardiac muscle biology: (1.) How do mesodermal cells become determined to enter the skeletal and cardiac muscle cell lineages? (2.) How are skeletal and cardiac muscle genes activated during development? (3.) How are quantitative modulations in gene expression controlled with respect to fast and slow skeletal muscle fiber types and cardiac muscles; and how are these controls modulated by the unique physiology of different anatomical muscles? (4.) How can the understanding of muscle gene regulation be developed into effective strategies for muscle gene therapy? (5.) What mechanisms regulate the mitogenic responsiveness of satellite cells during skeletal muscle regeneration? (6.) Can skeletal muscle cells and stem cells be modified so as to provide functionally beneficial replacements for injured skeletal and cardiac muscle?

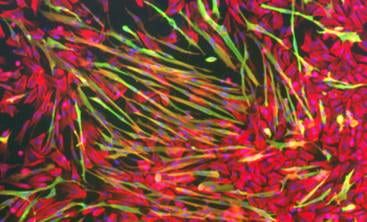
Early chick embryo cardiomyocytes can express endothelial cell proteins such as vonWillibrand’s Factor (Red), as well as muscle proteins such as myosin (Green).
These problems are studied in cell cultures, chick and mouse embryos, and transgenic mice. The systems are experimentally manipulated via cytokines, intracellular signal transduction pathways, transcription factors, and a diverse array of target gene regulatory regions. Studies of skeletal and cardiac muscle development focus on identifying cytokines and molecular mechanisms that induce myogenesis in chick embryo somites, and on inductive interactions that convert stem cells to cardiomyocytes.
Chick embryo somite following exposure to cytokines that induce skeletal muscle differentiation in some cells (Green), while others continue along alternative developmental pathways (Red).
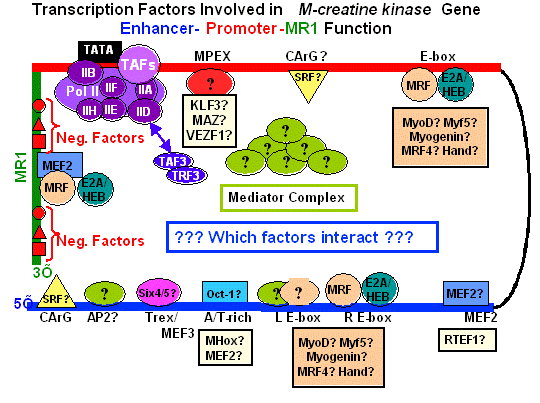
Studies of muscle gene regulation focus on identifying regulatory regions and control elements within muscle genes such as M-creatine kinase, and on transcription factors that interact with the identified elements. These studies involve quantitative proteomics to identify previously unknown DNA binding factors and ChIP analysis to identify factors bound to chromatin of cells in different developmental states. Related studies focus on the role of nuclear membrane components such as lamin and emerin in regulating muscle gene expression, and how mutations in these genes lead to muscle and cardiac diseases. Gene therapy studies concentrate on the design of regulatory gene cassettes that can be used for high-level expression of therapeutic proteins in human muscle diseases such as Duchenne Muscular Dystrophy, and cardiomyopathies, and on strategies for the external regulation of therapeutic gene expression. Muscle regeneration studies focus on using stem cells for constructing artificial muscles, and on determining the mechanisms that control when replicating muscle cells differentiate. Studies of heart muscle repair concentrate on deriving myogenic cell lines carrying gene modifications that potentiate the functional repair of experimental cardiac infarcts.
Publications: