Session 4: Drug Resistance & Second-Line Therapy
In session 3, we used the algorithm to manage clients based on the viral load test results. In this session, we’ll look at the final part of the algorithm: what to do for clients who have treatment failure (that is, switch to second-line therapy). We’ll also cover how ARV drug resistance affects clients. Note: much of the content presented this session is to provide a general understanding. It’s important that nurses refer to a mentor or doctor for clinical guidance.
Learning Objectives
By the end of this session you will be able to:
- Explain the challenges drug resistance plays on controlling HIV in a community and what countries are doing to minimize it.
- Describe the differences between primary, secondary, and cross resistances.
- List the risk factors for development of HIV drug resistance.
- Define treatment failure in adults living with HIV.
- Identify the challenges of switching a client to second-line therapy.
- Describe the process of switching a client to second-line therapy.
Learning Activities
-
Quiz: Pretest (10 min)
-
Reading: HIV Drug Resistance (15 min)
Drug resistance is a major problem that can happen while people are on treatment. If a client on ARVs is resistant to the drug, then the drug isn’t working to suppress the virus. Instead, the virus will continue to multiply, the CD4 cell count will decrease, and the client will show clinical signs of HIV disease progression.
Drug resistance makes it difficult to control HIV in a community. There’s a danger of transmitting a resistant virus to someone else, which in turn would limit the antiretroviral options that could be used to control that person’s virus. Resistance to drugs also decreases the effectiveness of ARV-based pre-exposure (PrEP) or post-exposure prophylaxis (PEP) or topical microbicides. PrEP failure due to pre-existing HIV resistance strains has been reported in the literature.
Additionally, underlying drug resistance results in more rapid treatment failure among people receiving first-line regimens and increases the need for second-line therapies. These therapies are more toxic, have more adverse events, are more difficult to adhere to, and cost more.
If we want to control the virus, we must take measures to avoid drug resistance developing in clients taking ARVs.
Unfortunately, over the years, more and more patients globally are developing resistance to ARVs. The 2017 HIV Drug Resistance Report, developed by WHO, CDC, and Global Fund to Fight AIDS, TB and Malaria, highlights a growing picture of increasing drug resistance that threatens the success of national HIV programmes. For Zimbabwe the pre-treatment drug resistance to efavirenz/nevaripine is >10%.
-
Reading: Types of Drug Resistance (10 min)
Tap each type of HIV drug resistance to read more.
Primary (or transmitted) resistance
Primary resistance occurs when ARV naïve clients (people who have never taken ARVs) are directly infected with a virus that’s already resistant to ARVs.
Secondary (or acquired) resistance
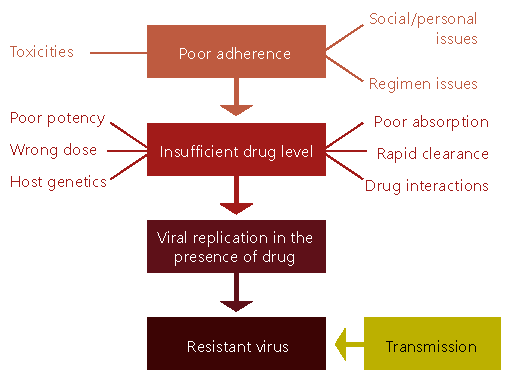
Secondary resistance is the most common type of resistance. It occurs when the virus continues to replicate even though the client is taking ARVs. This is caused by the level of drugs being too low to block viral replication, but high enough to exert a positive selection pressure on the virus.
This graphic explains how development of secondary drug resistance happens. As we talked about in session 3, there can be different reasons for poor adherence, such as social/personal issues, regimen issues, or toxicities, which lead to insufficient ARV blood levels. There are also many different causes for insufficient drug levels, which lead to the virus developing resistance.
Cross resistance
Cross resistance means that HIV has resistance to a medication in a drug class, such as the non-nucleoside reverse transcriptase inhibitor class (NNRTI), even though it’s never been directly exposed to that medication. This is a result of mutations that have been selected for by the use of another medication in that same drug class. Cross resistance is always limited to drugs within a class of antiretroviral agents. For example, if a virus has developed resistance to the NNRTI nevirapine, it will already be resistant to efavirenz. Continuing on a failing ART regimen encourages the development of cross resistance.
-
Reading: Risk Factors (10 min)
Let’s look at the risk factors for drug resistance. Tap each factor to read more about it.
Virus
The virus itself makes mistakes during replication and can cause resistance to drugs. There are two basic terms we use when talking about resistance. A wild type virus is a virus without any resistance mutations. It has no resistance to drugs. A mutant virus has changed its structure, leading to drug resistance.
HIV replication is a highly error-prone process. With HIV, drug resistance is caused by changes (mutations) in the virus's genetic structure. These mutations can lead to changes in certain proteins, most commonly enzymes, which help HIV reproduce (replicate).
Mutations are very common in HIV. This is because HIV replicates at an extremely rapid rate and doesn’t contain the proteins needed to correct the mistakes it makes during copying.
Mutations occur randomly on a daily basis, but many are harmless. In fact, some mutations actually put HIV at a disadvantage—they reduce the virus's "fitness" (called viral fitness) and slow its ability to infect CD4 cells in the body. The virus’s ability to replicate may be impaired, and it may be more susceptible to other drugs.
For example, the M184V mutation causes resistance to lamuvidine (3TC). Lamuvidine is kept in the second-line therapy even though the virus is resistant to it because the M184V mutations makes the virus more susceptible to other drugs used in second-line therapy.
A number of mutations can actually give HIV a survival advantage when HIV medications are used, because these mutations can block drugs from working against the HIV enzymes they are designed to target. These are the mutations we are concerned about when we talk about drug resistance.
Host
The host is the person who’s infected with the virus. Resistance can develop
- When the person (host) doesn’t take all of the ARVs prescribed at the right time and the correct amount every day
- If the person has a condition that affects the absorption of the medication (such as chronic diarrhoea or vomiting)
- If the person is taking other drugs that interact with ARVs
Treatment
Some drugs require only one mutation (or mistake) to develop resistance, for example lamivudine. Other drugs require the virus to have more mutations before resistance develops (for example, protease inhibitors). These are said to have a high genetic barrier to resistance.
-
Quiz: Knowledge Check (10 min)
-
Reading: Treatment Failure (10 min)
Treatment failure occurs when the HIV infection is no longer controlled by an ARV regimen, such as with drug resistance.
The virus can develop resistance if the level of drugs is insufficient to block replication, but can exert a positive selective pressure on variants with decreased drug susceptibility. Resistance is most often a consequence, not the cause, of initial treatment failure.
A client must be on ART for at least six months with good adherence before we can consider treatment failure. If a client becomes unwell during these first six months on ART, he or she may be experiencing immune reconstitution inflammatory syndrome (IRIS) or a new opportunistic infection (OI).
Treatment failure can be either virologic failure, immunologic failure, clinical failure, or a combination of the three. Tap on each type of failure to learn about its definition for adults and how to assess failure (as defined by the WHO Consolidated Guidelines, June 2013).
Virologic failure
Virologic failure results when the viral load isn’t completely suppressed, leading to resistance. Virologic failure will be noticeable before immunologic or clinical failure.
Definition: Viral load 1000 copies/ml and above based on two consecutive viral load measurements after three months of enhanced adherence counselling (EAC) and good adherence.
Assessment: Viral load tests
Immunologic failure
Definition: CD4 cell count falls to the baseline (or below) or persistent CD4 levels below 100 cell/mm3
Assessment: CD4 cell count
Clinical failure
Clinical failure is the last of the three types of treatment failure to become evident. At this point, the viral load will have already increased and the CD4 count will have dropped.
Definition: A new or recurrent clinical event indicating severe immunodeficiency (WHO stage 4), such as cryptococcal meningitis.
Assessment: Assess clients for new or worsening clinical symptoms or the development of an opportunistic infection or malignancy after a person has been on ART for six months.
When suspecting clinical or immunological issues, it’s important to get a targeted viral load test to make sure that the client doesn’t decline further.
-
Quiz: Knowledge Check (5 min)
-
Reading: Why Use Triple Therapy (5 min)
When we start our clients on ART, we give them a combination of three drugs (known as HAART). Effective combination therapy can block drug resistance, since more than one mechanism (virus, host, or treatment) would have to be compromised for resistance to develop for all drugs in the regimen.
Early research in HIV disease management showed that giving single or double drug therapy wasn’t effective in controlling the virus. Triple therapy proved more effective at suppressing viral replication, which is why it continues to be the standard treatment for HIV disease. If the virus can’t multiply in the body, it can’t create new mutations because mutations develop by chance, so the probability will be low if there is minimal replication.
-
Reading: Second-Line Therapy (10 min)
Now let’s talk about what to do once a client has been diagnosed with treatment failure. In the algorithm, this is the final step for clients with unsuppressed or high VL. Clients whose HIV hasn’t been successfully controlled on the first-line therapy will need to be treated with a different regimen. The second-line regimen will still include two NRTIs, such as ABC and 3TC, and then add a PI such as ATZ/r or LPV/r.
A thorough adherence assessment and the timing of the switch is critical. Tap on each issue to read more.
Adherence
If we switch a client who has poor adherence (because of mental health challenges or substance abuse) from first-line to second-line therapy, we risk that client quickly developing resistance to the new regimen.
Timing
Clients switched too early may unnecessarily experience side effects and could potentially jeopardize our ability to effectively treat HIV with first-line regimens. Switching a client too late puts him or her at risk of becoming sick or developing an OI due to a weak immune system. The effectiveness of the second-line therapy is also at risk.
As the viral load increases, more mutations accumulate. The more mutations there are, the more likely a client will have pan-resistance (resistance to all medications). So if a client continues for a long time on a failing regimen, then he or she will accumulate more mutations. If a client is switched to a different regimen early once experiencing treatment failure, then there’s a better chance that the second-line regimen will work.
While second-line therapy is a viable option for treatment failure, there are issues that go hand in hand with switching a client. Pill burden, adherence issues, difficulty accessing the medications, increased costs for medication, higher adverse effects, and costly laboratory monitoring are just a few of the challenges. For these reasons, we should try to keep patients on first-line therapy for as long as possible while conducting enhanced adherence counselling. Adherence issues must be completely ruled out before switching to second-line therapy. Enhanced adherence counselling and viral load monitoring are essential for preventing unnecessary switching of treatment regimens.
-
Reading: Second-Line Therapy Process (10 min)
Tap on each step to read about the process of switching clients to second-line therapy. This should always be initiated in consultation with a specialist in HIV and AIDS treatment or with the clinical mentorship team at the OI/ART clinic.

Step 1
Before switching a client to second-line therapy, order a viral load test.
Step 2
The client should undergo a full examination that includes reviewing and updating the medical history, laboratory testing for glucose and lipid levels, TB screening, and EAC.
Step 3
Once the tests indicate that second-line therapy is appropriate, switch the client to the new therapy. If the client was previously on tenofovir disoproxil fumarate (TDF), check for Hepatitis B serology. If the client tests positive for HBsAG, TDF must be continued in the second-line regimen as it is a treatment for Hepatitis B.
Step 4
After the client starts second-line therapy, the number of visits increases. Once the client is stable, he or she can move into appropriate differentiated care.
-
Quiz: Steps for Switching (5 min)
-
Reading: Selecting Drug Regimen (10 min)
Here’s a table showing preferred second-line therapies for specific populations.
Target population Preferred second-line regimens Adolescents (10-19 years old), adults, and pregnant and breastfeeding women If TDF was used in first line ART AZT+3TC+ATZ/r or LPV/r If AZT was used in first line ART TDF +3TC +ATZ/r or LPV/r HIV and TB co-infection Patients receiving Rifampicin Same NRTI backbone as recommended for adults and adolescents plus double dose LPV/r (800mg/200mgBD) HIV and HBV co-infection AZT + TDF +3TC +ATZ/r or LPV/r* -
Reading: Early Warning Indicators (10 min)
HIV drug resistance is a major threat for countries scaling up ART programmes. The WHO has created early warning indicators (EWIs) of HIV drug resistance (HIVDR) to minimize HIVDR in these countries. EWIs are quality-of-care indicators that assess factors at ART clinics experiencing HIVDR. In Zimbabwe, the purpose of collecting EWIs for HIV drug resistance is to assess the extent to which the ART program is functioning to optimise prevention of HIVDR.
The EWIs are a package of eight indicators, one of which is viral load suppression at 12 months. This indicator should be measured only at facilities where routine viral load monitoring is performed for all patients 12 months after ART initiation.
Here are the eight indicators:
EWI EWI taget (%) Percentage of initial ART prescriptions congruent with national/WHO guidelines 100 Percentage of clients lost to follow-up at 12 months ≤20 Percentage of clients retained on first-line ART at 12 months ≤70 Percentage of clients with 100% on-time drug pick ups during the first 12 months of ART (or some other specified period) ≥90 Percentage of clients who attend all appointments on time during the first 12 months of ART (or some other specified period) ≥80 Percentage of clinics with ARV supply continuity during a 12-month period 100 Percentage of client adherence to ART by pill count or some other standardised measure ≥90 Percentage of clients with viral load < 1000 copies/mL at 12 months ≥70 Data on the indicators comes from routine collection of medical and pharmacy records, specifically monitoring for factors associated with HIVDR prevention or emergence.
If a facility doesn’t achieve the indicator targets, drug resistance won’t necessarily result immediately. However, meeting these targets will help minimise preventable HIVDR. Sites that don’t meet one or more EWI targets may require increased resources, staff training, or additional review to clarify the kind of support needed to be able to meet these targets.
-
Reading: Patient Education (10 min)
It’s important to educate clients about about signs and symptoms of possible treatment failure. They should know that weight loss, recurrent infections, new rashes, or any new staging condition could be indicators of treatment failure. If they experience any of these symptoms, they should report them promptly to their health facility. This education will help encourage clients with possible treatment failure to attend the facility before a booked appointment.
Of equal importance, we must prepare clients for starting second-line therapy (much as in the way we prepare them for first-line therapy). As we’ve learned in this session, clients moving to second-line therapy often have had poor adherence, so we want to make sure that they start the second-line regimen with excellent adherence.
The OSDM provides guidance for counselling and preparing clients switching to second-line therapy (pp 84-85).
-
Case Study: Isaac (15 min)
In session 1, we met Isaac, a 39-year-old farmer who tested positive for HIV. In session 2, we went through the visit where he had a VL test. In session 3, we saw his results.
Isaac has been seen in the clinic monthly since his viral load results. At each visit, he has returned his pill container empty and reported no missed doses.
He has seemed more withdrawn at each visit and only speaks when asked direct questions. He has consistently reported drinking only a few beers occasionally. When asked about Rumbi, he replies that “She’s good.”
Isaac’s viral load was repeated three months after the last test and the result is 6, 900 copies/mL.
Isaac asks you if having a high VL means he’s going to get sick and die. He’s visibly upset and tells you that he needs to stay healthy to work the farm and care for Rumbi. He’ll do whatever you say to make the number go down.
After explaining what the high VL means to Isaac, he tells you that he's very worried. He stopped his medication several weeks ago since he felt better. When he was taking the pills, he had trouble waking up early to feed his animals and felt tired most of the morning when he was tending his field. When he was taking the pills, he had trouble waking up early to feed his animals and felt tired most of the morning when he was tending his field. Since that did not happen, he thought that it was OK to continue to not take the pills. He hasn’t told Rumbi any of this and wishes that you don’t tell anyone. He tells you that since the test result is worse than the last one, he’ll start taking the medicine and promises not to stop again.
You consult with the doctor and explain Isaac’s case. It’s decided that Isaac can resume his original ART regimen of EFV/3TC/TDF with enhanced adherence counseling. If adherence is good, the VL test will be repeated after three months. His viral load should decrease by at least 1 log or be undetectable. If not, a switch to second-line therapy will be needed.
Over the next three months, Isaac attended two clinic appointments on time. At the third appointment, he came a day early because he was taking a trip with Rumbi to visit her family. When asked about his alcohol consumption, he said that he drank only one or two beers on the weekend and that he felt much better. He reported that the tired feeling he was having seemed to go away, which he attributed to drinking less. Isaac was dispensed a 30-day supply of EFV/TDF/3TC at each visit. He brought his pill container to each visit. Below is the pill count for the three months of enhanced adherence counseling:
- EAC visit 1: 2 pills remaining
- EAC visit 2: 3 pills remaining
- EAC visit 3: 0 pills remaining
After three months of EAC, Isaac’s viral load is tested. He brings Rumbi with him to hear the result. They are both pleased that it’s 496 copies/mL. Isaac reports that over the past several months he was very worried that he was going to get sick and die. He admitted that he was angry and ashamed of being HIV positive and also worried that Rumbi would leave him. He promises that he will continue to take his medication.
-
Case Study: Rachel (15 min)
Rachel is 24 years old and has been living with HIV for three years. She was diagnosed during a hospital stay for severe pneumonia, which was later diagnosed as PCP. She also had oropharyngeal and esophageal candidiasis and anaemia. Her past medical history included probable herpes zoster when she was 19 years old.
Rachel’s baseline laboratory results while in the hospital were:
- CD4 cell count: 96 mm3
- Haemoglobin: 8.6 g/dL
After her PCP and candidiasis were successfully treated, Rachel began ABC/3TC/NVP. At her two-month follow-up appointment it was discovered that Rachel had only been taking one NVP pill daily rather than one pill twice daily. She told the clinic nurse and pharmacist that she thought that was the correct way to take the pills. Rachel increased her NVP without any side effects.
Her haemoglobin continued to be below normal for several months. After about a year on ARVs, Rachel was tested again. This time her results were:
- CD4 cell count: 222 mm3
- Haemoglobin: 11.0g/dL
Approximately two years after starting ARVs, Rachel’s CD4 cell count was 310 mm3. She continues on ABC/3TC/NVP and cotrimoxazole.
Rachel has come to the clinic today asking for a pregnancy test. She has been with her boyfriend for about a year and she disclosed her HIV status to him soon after they met. He tested negative after she told him but hasn’t been tested since that time. Her pregnancy test is negative, but Rachel is visibly upset. She tells you that she was hoping to get pregnant and that her boyfriend has told her he wants a child. She’s afraid that if she can’t become pregnant he will leave her. You’re concerned not only about what Rachel has shared but also by her appearance. You’ve known her for many years and today she looks sick.
Physical examination reveals oral candidiasis and enlarged, tender cervical and axillary lymph nodes. She tells you that she hasn’t been feeling well, but thought it was because she had started a new job that required her to stand for most of the day. Recently she has had a sore throat and trouble swallowing, but thought she had caught a cold from her boyfriend. The sore throat has made it difficult to eat, so she thinks she has lost weight (less than 10%). She’s able to drink water and tea.
Further discussion with Rachel reveals that she stopped taking her ARVs a year ago even though she filled her prescriptions every month and told the staff that she had not missed any doses. She begins crying and tells you that she was tired of taking pills and she felt better when she wasn’t taking them. When she thought she was pregnant she started taking them again a few weeks ago but she hasn’t felt well, has a poor appetite, was bothered by the “stuff” in her mouth, and had trouble swallowing. She knew that she needed to take the pills to protect her baby.
You draw blood for a viral load test and prescribe fluconazole. You instruct her to continue taking her current ARVs until the results of her viral load test is back. She’s given an appointment to return to the clinic in one week or sooner if her symptoms worsen.
Results of her laboratory tests are as follows:
- Haemoglobin: 11.2 g/dL
- Viral load: 51,060 copies/mL
Rachel’s candidiasis resolved completely and her appetite improved, resulting in her feeling much better and gaining weight. She reported no missed doses during the month, although she took the medication an hour or so earlier than usual a couple of nights.
Rachel’s repeat viral load result after three months of enhanced adherence counselling and improved adherence was 4,100 copies/mL.
Rachel’s very upset. She came to this visit with her boyfriend Tendai who was outside the examination room. He comes into the room when he hears her crying. She tells her Tendai that the medicine she has been taking stopped working and she’s afraid she’s going to die. Tendai asks you to explain what is going on. He doesn’t understand since Rachel has been taking the medicine for many years.
Tendai tells you that he has been reading about HIV and the treatments that are available. He’s worried about the number of pills Rachel will have to take if her medication is changed and asks you why she can’t take the medication that is only one pill once a day. He knows of many other HIV-positive people that are taking that pill and they are very healthy.
Rachel’s ART regimen was changed to TDF/3TC/ATZ/r
Rachel was concerned with the number of pills she had to take and also that the medication had to be taken with food. Although her appetite improved once the candidiasis cleared up and she wasn’t having any nausea, Rachel was still not eating well. Tendai tried to comfort Rachel and promised that he would help her as much as possible. A follow-up appointment was scheduled for one month. Rachel was given 30 ATZ/r, 30 TDF/3TC.
Rachel and Tendai returns to the clinic two days earlier than her appointment due to a conflict with their work. They are smiling and telling you that Rachel is feeling much better. She proudly reports that she has been taking the new medication every day without any trouble and hasn’t missed any doses. She returns her pill containers with the following amounts: One ATZ/r and one TDF/3TC.
-
Reading: Summary (5 min)
This concludes session 4. We discussed causes of drug resistance and the challenges it plays on controlling HIV. We then drilled deeper into this topic by examining the three types of drug resistance — primary, secondary, and cross resistance. Next, we learned about the risk factors for drug resistance. Moving onto treatment failure, we learned about causes for treatment failure and how the WHO defines treatment failure for adults. We then talked about triple therapy and the process to take to successfully switch a client to second-line therapy. We also learned about the WHO’s early warning indicators used to assess HIV drug resistance in countries scaling up ART programs. Last, we wrapped up with a discussion of patient education, focussing on educating clients about signs of treatment failure and starting second-line therapy.
-
Quiz: Posttest (10 min)