Session 5: VL Monitoring for Pregnant and Breastfeeding Women, Children and Adolescents
This session will focus special populations. We'll talk about the importance of viral load monitoring for HIV-positive pregnant and breastfeeding women and review how VL monitoring for pregnant and breastfeeding women differs from the algorithm used for adults. We'll also look at issues related to children and adolescents with high VL, focussing on how treatment failure is defined for this age group.
Learning Objectives
By the end of this session you will be able to:
- Explain the importance of viral load monitoring for pregnant and breastfeeding women.
- Describe the algorithm used for viral load monitoring for pregnant and breastfeeding women.
- Define treatment failure for children.
- Describe factors to consider when a child has a high viral load.
Learning Activities
-
Quiz: Pretest (10 min)
-
Reading: Prevention of Mother-to-Child Transmission (PMTCT) (5 min)
Worldwide, the most successful intervention in the HIV epidemic is the prevention of mother-to-child transmission (PMTCT). Stopping mother-to-child transmission not only reduces infant mortality, but it also is seen as a first line of defense against the spread of the epidemic.
PMTCT is a social justice issue since without the proper care and treatment most babies infected with HIV will die in the first two years of life. Hindering this transmission is also cost effective as it will save on the need for future HIV treatment.
HIV can be passed on from a mother to her baby during pregnancy, during delivery, and while breastfeeding. Without ART or other interventions, the likelihood of mother-to-child transmission is between 15% to 45%. This percentage varies depending on whether the mother is exclusively breastfeeding or not. Without breastfeeding, there is a 15%-30% risk of transmission to the baby. With breastfeeding, this risk is slightly higher at 25%-45%. Due to the blood contact during delivery there is a high risk of transmission. For this reason, most of the infections tend to occur during labour and delivery.
-
Reading: Elimination of Mother-to-Child Transmission (eMTCT) of HIV and Syphilis (15 min)
Recently, there has been a shift in focus from prevention of mother-to-child transmission to elimination of mother-to-child transmission.
To promote the goal of eliminating mother-to-child transmission of HIV and syphilis, the WHO has published a report titled Elimination of mother-to-child transmission (EMTCT) of HIV and syphilis: Global guidance on criteria and processes for validation that provides global guidance. In the report, they establish eMTCT validation targets and indicators that can be used by national, regional, and global committees and organizations to monitor progress and validate countries who have successfully eliminated HIV and/or syphilis.
Tap on the following boxes to see the targets established by the WHO for HIV and syphilis elimination:
HIV
Impact criteria Process criteria - MTCT < 2% non breastfeeding populations or < 5% in breastfeeding populations
- Case rate ≤ 50 per 100,000 live births
- ANC coverage ≥ 95%
- Testing coverage ≥ 95%
- ART coverage ≥95%
Syphilis
Impact criteria Process criteria - Case rate ≤ 50 per 100,000 live births
- ANC coverage ≥ 95%
- Testing coverage ≥ 95%
- ART coverage ≥ 95%
High levels of coverage combined with high quality services have enabled some countries to reach these targets and achieve true elimination of mother-to-child transmission of HIV and syphilis. In 2015, Cuba was the first country to be validated by the World Health Organization. Since then, Thailand, Belarus, Armenia, and Moldova have also received validation.
However, in Sub-Saharan Africa, which account for 90% of the global disease burden, these targets haven’t been reached. This doesn’t mean that countries in Sub-Saharan Africa haven’t made progress. Quite the opposite, actually. But the prevalence of HIV is so high in many of these countries that even with very successful eMTCT programmes, it’s unlikely that they will meet the paediatric incidence criterion of 0.05%, or 50 cases per 100,000 live births.
With ART and other interventions, transmission can be reduced to less than 5% in developing countries and to less than 2% in developed countries. Both globally and nationally there is a four-pronged comprehensive approach to eliminating mother-to-child transmission that has been widely accepted. Tap on each of the prongs to learn more:
Prong 1: Primary HIV prevention
Focuses on keeping women and men HIV negative through:
- Provision of access to condoms
- Provision of early diagnosis and treatment of STIs
- Making HIV testing and counselling widely available including retesting for women during pregnancy and when breastfeeding. HIV-negative women should be tested during their first trimester, third trimester (or at delivery), six weeks postnatal, and every six months while breastfeeding.
- Provision of suitable counselling for women and men who are HIV negative
Prong 2: Prevention of unintended pregnancies among women who are HIV-infected
Focuses on reproductive choices and family planning for people living with HIV:
- Appropriate support so that women who know they are HIV infected can avoid unintended pregnancies and therefore reduce the number of infants at risk for mother-to-child transmission
- Access to counselling and testing services
- Effective family planning to help prevent unintended pregnancies
- Dual protection (use of male or female condom and another family planning method)
- High quality reproductive health counselling to help clients make informed decision making about pregnancy choices
Prong 3: Prevention of HIV transmission from HIV-infected women to their infants
Addresses care for women during pregnancy, labour, and delivery and thereafter including their infants:
- Antiretroviral treatment and prophylaxis
- Safer delivery practices
- Safer infant and young child feeding practices
Prong 4: Provision of treatment, care, and support to women living with HIV and their infants, partners, and families
Provide support to women living with HIV to ensure that they remain adherent to ART and retain health care throughout pregnancy, breastfeeding, and for life. Specific avenues of support include:
- ART for all HIV-positive pregnant women for their own health
- Care and support for HIV-exposed infants
- Nutritional support for HIV-exposed infants
- Treatment, care, and support services for women and their families
-
Reading: PMTCT Testing and Services (10 min)
Let’s briefly review when to test pregnant and lactating women for HIV:
- At the first trimester of pregnancy or at first contact,
- At third trimester or at delivery (32 to 34 weeks gestation),
- Six weeks post-natal, and
- Every six months during breastfeeding.
If the test is positive, evaluate the client by getting a baseline CD4 cell count (which isn’t an indicator for treatment), assessing renal function (with a urine dipstick or creatinine level), testing the client’s haemoglobin, taking the client’s history, and conducting a clinical exam.
Under treat all, every pregnant woman is eligible for ART if she tests positive for HIV. It’s important to prepare clients psychologically for life-long ART. You should also provide them with routine pregnancy-related care as well as clinical, immunological, and virological assessment and review.
If you need to refresh your skills on treating HIV-positive pregnant women, go through the PMTCT Client Retention training on this tablet.
The Consolidated HIV & AIDS Job Aide, page 88 (in the resouces), has information on re-testing for pregnant and lactating women.
It’s also important that we know which mothers are at high risk of transmitting HIV to their babies. The risk for mother-to-child transmission is higher
- If the mother’s VL is over 1000 copies/mL at 32 weeks (or more) gestation
- If the mother is newly diagnosed with HIV during labour and delivery or while breastfeeding
- If the mother isn’t on ART (or has less than eight weeks of ART when the baby is delivered)
The risk is low if these risk criteria aren’t met.
-
Quiz: Knowledge Check (5 min)
-
Reading: VL Algorithm (10 min)
In terms of mother-to-child transmission, the higher the viral load of the mother, the higher the risk of transmission to the baby. For this reason, HIV-positive pregnant and breastfeeding women should be prioritised for early initiation on ART and for viral load monitoring.
The algorithm for HIV-positive pregnant women and breastfeeding mothers is different than the one for the general population infected with HIV. This algorithm further divides this special population into women already undergoing treatment and women who are newly diagnosed or newly on treatment. For each group, tap to see how the algorithm differs.
Pregnant Women on ART
Pregnant women on ART should receive a VL test during their first ANC visit. The viral load should then be reviewed within two weeks.
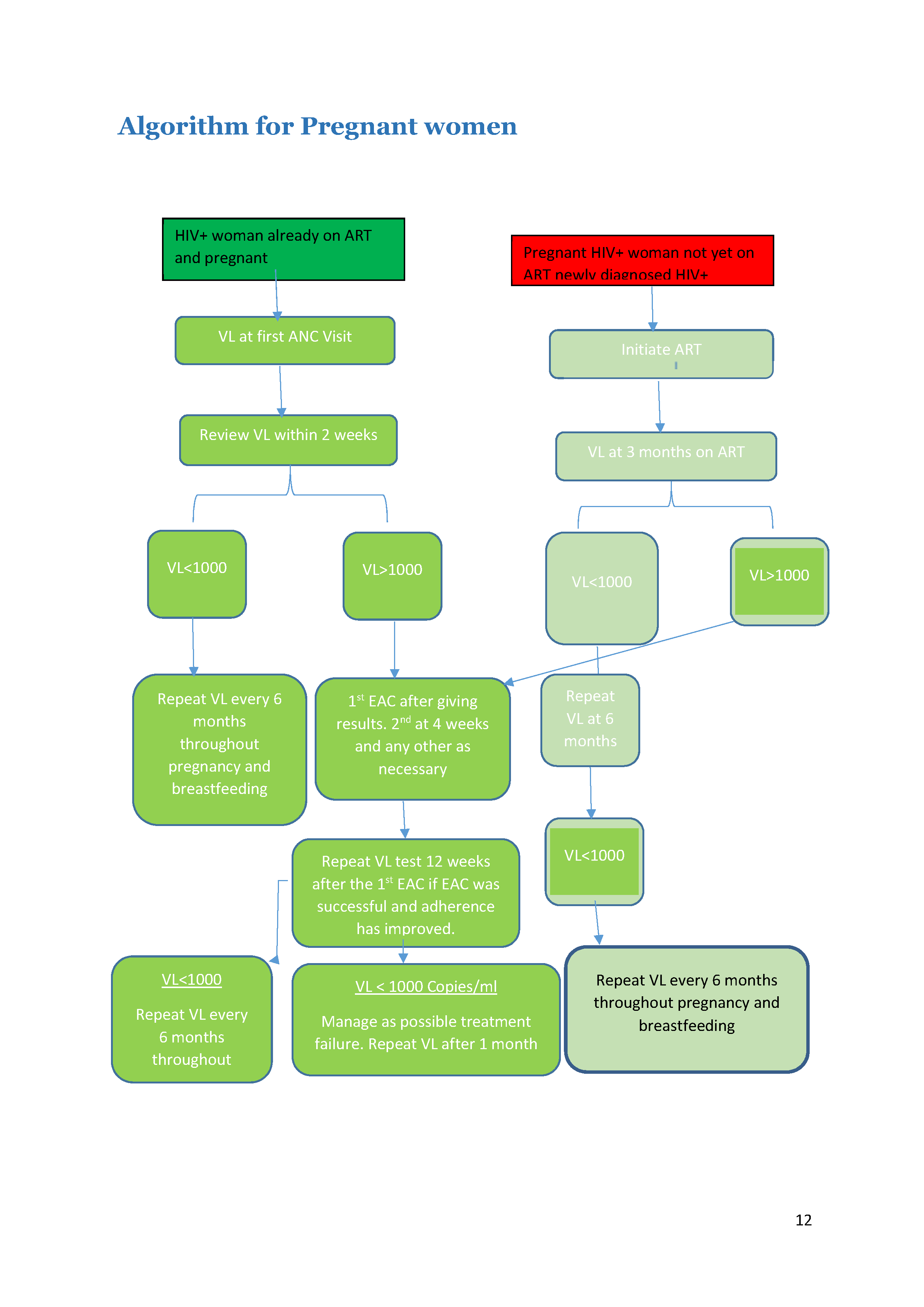
Pregnant HIV+ women not on ART
First, women who test positive should get a confirmatory test and start ART immediately. After three months on ART, conduct a VL test.
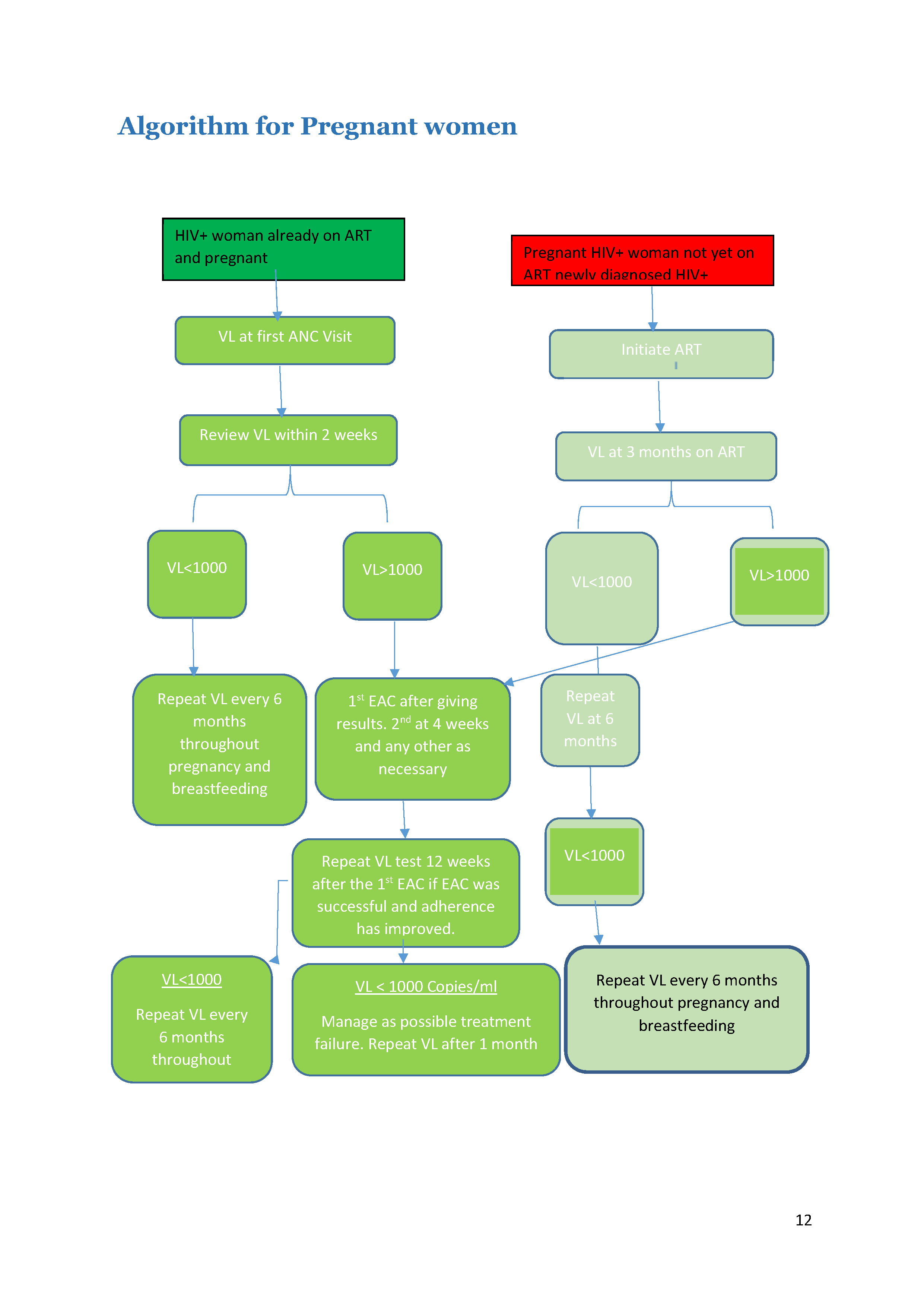
The rest of the algorithm is the same for each group. Tap on each category to see how to manage clients with high or low VL.
Low VL (< 1000 Copies/mL)
If the client has a low VL (< 1000 copies/mL), then the test should be repeated every six months throughout the remainder of pregnancy and breastfeeding.
High VL (> 1000 Copies/mL)
If the client has a high VL (> 1000 copies/ml), then enhanced adherence counselling (EAC) should be offered after giving the results to the client. Because the goal is to prevent mother-to-child transmission, for pregnant and breastfeeding women we need to assume drug resistance is the cause for high VL earlier than we would for the general population. Therefore, if the second VL result after EAC shows a high VL, we would assume drug resistance has developed and would switch the client to second-line therapy.
-
Case Study: Celesile (15 min)
Celesile is 22 years old. She tested positive for HIV today during her first ANC appointment. She’s six months pregnant. Her family doesn’t know she’s at the clinic since they believe in traditional birth attendants for pregnancies unless there’s a complication. The father of her baby left the village to go work with a long-haul trucking company. He wasn’t happy when he found out Celesile was pregnant, and she hasn’t heard from him in several months.
This is Celesile’s first pregnancy. She reports no problems, but felt that she wanted a nurse to check her baby to make sure everything was fine. Her past medical history includes treatment at another clinic for an STI and a painful rash across her abdomen, for which she was given medication. Both problems cleared up. She doesn’t remember what the names of the pills were, but one of them she took for over a week.
Vital signs and physical examination findings are as follows:
- B/P: 120/74
- Pulse: 70 and regular
- RR: 16
- Temperature: 37 ℃
- Height: 152 cm
- Weight: 57 kg
- HEENT – WNL: no thrush noted
- Chest: lungs clear to auscultation
- Breasts: no abnormalities noted
- Skin discolored and scarred across the rights side of the abdomen from the umbilicus to the right hip area. Celesile tells you that this is a result of the rash she had.
- Musculoskeletal: no LE edema noted
- Foetal heart rate: 132 beats per minute
- MUAC: 24
Your psychosocial assessment reveals that Celesile doesn’t drink alcohol or smoke. She admits to feeling “sad” at times and worries about raising her baby without her boyfriend and about her mother and grandmother finding out she’s HIV positive.
Laboratory findings:
- Hb: 8.2 grams per deciliter
- Urine glucose/protein: negative
- CD4 cell count: 320 cells/mm3
- RPR: negative
Differential diagnoses:
- HIV-positive asymptomatic
- Scarring secondary to probable herpes zoster
- WHO clinical stage 2
- P-0 G-1
Plan:
- EFV+3TC+TDF = FDC 1 tablet daily at bedtime beginning today
- Cotrimoxazole 960mg daily
- Folate, iron, and multivitamin daily
Celesile returns to the clinic at 35 weeks. She has been taking her medication every night and reported no missed doses. Initially, she complained of feeling very dizzy and tired immediately after taking the FDC but that feeling went away after a few weeks. She’s anxious to have her blood test today to see how well the medicines are working and tells you that she’s worried about her baby becoming infected.
Celesile’s viral load result is 543 copies/mL. When you contact Celesile, she's very happy and tells you that she’s planning on delivering her baby at home with the birth attendant. Her mother and grandmother have gotten upset every time she talks about going to a hospital or clinic to have her baby, so she doesn’t have a choice. She understands that she needs to stay on the medicine and exclusively breastfeed her baby.
Celesile’s labour didn’t progress and she was taken to the hospital by her mother and grandmother. She delivered a healthy baby girl weighing 2.8 kg. The baby was initiated on NVP prophylaxis and cotrimoxazole at six weeks. The PCR at six weeks was negative. Celesile still refused to disclose her HIV status to her family.
At her six-month visit, Celesile complained about fatigue and facing challenges breastfeeding because of her work schedule.
When asked about her adherence to her ARVs, she admitted to missing a few doses when she fell asleep before taking the pill. Adherence was documented in her record as “good”, but two entries were listed as “fair”. A pill count wasn’t done since she didn’t bring back the container.
Celesile’s viral load result was 856 copies/mL. She was continued on her regimen of EFV/3TC/TDF.
Further discussion with Celesile revealed that there were times when she woke up around 2:00 am and thought about taking her medication, but decided that it was too late and went back to sleep. She asks you what you think she should do if this happens again.
Over the next several months, Celesile reported better adherence but continued to have occasions where she would fall asleep before taking her medication. She would take the medication when she woke up in the middle of the night, but this caused dizziness and sleepiness, which affected her work.
On two occasions she forgot to bring her pill container, but overall, her adherence was documented as ranging from 91 – 96%. She discussed beginning formula feeding, but her mother convinced her to continue breastfeeding at least until her daughter was one year old.
At her 12-month clinic visit, Celesile reported that she was going to stop breastfeeding over the next month. The lab results from her 12-month visit are:
- CD4 cell count: 382 cells/mm3
- Viral load: 640 copies/mL
At that visit, Celesile asked to have her baby tested, She was upset when she learned it couldn’t be done.
Celesile’s daughter was tested and the result was negative.
During one of her refill visits, Celesile requested to see the nurse. She was upset because she heard that if the viral load test result was a number and not the letters TND, then the medication was not working properly.
-
Case Study: Hazel (15 min)
Hazel is 37 years old and presents to the ANC clinic asking for a pregnancy test. She tells you that she hasn’t had regular menstrual cycles for the past several years, but her friend told her to come today since she told Hazel that she “looked pregnant”. Hazel lives with her sister and her family; their parents are deceased. Hazel is recently divorced and has no children.
Hazel’s past medical history includes treatment for an STI many years ago, but Hazel doesn’t recall what medication she was given. She reports that she finished all of the pills. Hazel also reports having a painful rash on her stomach two years ago. She went to a local clinic near where she was working and took pills until the rash disappeared.
Hazel’s pregnancy test is positive and after further discussion about her menstrual cycles and the date of her last period, it’s estimated that Hazel is 35 or 36 weeks pregnant. She tells you that she and her ex-husband tried to get back together last spring. She thought he was happy being back with her but one day he came home and told her that he wanted a divorce and decided to take a job he was offered in Zambia. She hasn’t seen or heard from him since he left.
Hazel didn’t react when told about the pregnancy test results and remained very quiet throughout the visit and during the physical examination. When asked if she felt any movement from her baby she replied that she “thought it was gas.” When asked about a prior HIV test, Hazel tells you she never had one. You explain the importance of knowing her HIV status for her health and her baby’s health. Again, her affect is very dull. She tells you to “do whatever needs to be done”.
Hazel’s HIV test is positive. She becomes angry when she hears the result. “I had a feeling he was fooling around. I should’ve known he would try to hurt me.” When you ask her about this statement she tells you “It doesn’t matter anymore. I’m going to die and who will take care of this child, who probably has the virus too”?
Throughout the discussion about HIV, its treatment, and the medication she will be giving her baby, Hazel remains quiet, answering only when asked specific questions. She tells you that she understands how to take her medications: EFV/TDF/3TC=FDC once daily and cotrimoxazole 800/160 once daily (Hazel lives in an area where malaria is highly prevalent). She doesn’t have any questions about the schedule or the medication’s possible side effects.
You ask about disclosing her status to her ex-husband, sister, or another person who will support to her. She replies, “I’m fine and no one needs to know about this. My husband’s gone and I want nothing to do with him. My sister and her family wouldn’t let me live with them if they knew this!” You’re feeling uneasy about Hazel’s situation and her reaction to the news, so you ask her to return to clinic in one week. She agrees.
Before she leaves the room, Hazel asks you several questions that you know you’ve already addressed when you provided patient education. She asks if she needs to start taking the medication today, why she can’t take the pills in the morning rather than at night, and if it would be better to take two pills so the medication works stronger in her body. She then asks you if the medication will keep her baby healthy, or is it too late? You repeat the information about how to take the medication and reinforce the advantages and importance of disclosing her status to either a family member or friend who will be a support to her. Again, she refuses.
Hazel returns to the clinic as directed. She reports feeling very tired in the morning, lasting until mid-morning. She reports no missed doses, but forgot to bring her pill containers back. She asks you again about her baby having the virus and begins to cry. “How am I going to know if the medicine is working for my baby? I’m not worried about myself, I’m worried about this baby!”
Hazel asks you to explain why breastfeeding is recommended rather than formula feeding. She doesn’t understand how the virus won’t get into her baby through breast milk, but she’s also afraid that if she gives the baby formula, her sister will want to know what’s wrong.
Over the next few weeks, Hazel comes back to the clinic for unscheduled visits primarily due to continuing mild to moderate side effects of EFV. Most bothersome are the vivid dreams and restless sleep. Her difficulty with sleep is also a direct result of her not being able to find a comfortable sleeping position and indigestion. She continues to report no missed doses.
At 39 weeks, Hazel’s labour begins. She delivers a healthy baby boy weighing 3.3 kg without complications. The birth PCR was negative.
Her sister is with her during the birth and delivery and asks Hazel about the medication the baby needs to take. Hazel has told the staff not to say anything about her HIV. She tells her sister the medication is needed to help the baby’s blood. There seems to be quite a bit of tension between Hazel and her sister. Hazel mentions to her sister that she is thinking about formula feeding her son, since she wants to find a job to help with expenses. Her sister tells Hazel that formula isn’t good for a baby and only breastmilk can keep her baby healthy!
Hazel asks if she can have her baby tested again for the virus since she is worried that the test may have been wrong.
Hazel continues to report no missed doses and that she’s not as fatigued as she was when she first started the medication. The viral load test taken after she’d been on ARVs for approximately three months is 2210 copies/mL.
-
Reading: VL Monitoring for Children (10 min)
When monitoring the VL for children and adolescents, follow the algorithm for the general population with HIV.
Children with High Viral Loads
There are some different actions you can take with children with high VL. According to the OSDM,
...children and adolescents have higher rates of treatment failure than adults. Children are usually dependant on a caretaker for administration of their ARVs and, for many who are orphaned, that caretaker often changes or is elderly. When a child is identified with a high viral load, the multidisciplinary team at the clinic should discuss this case and consider a home visit. An assessment by a social worker may also be needed. Investigating further community support for this child should be considered. There may be another adult or peer on ART living in the same community who may be willing to support the child and their family as a daily treatment buddy to improve adherence to medication. Another common barrier to adherence for children and young adolescents is non-disclosure. Working with the caretaker to fully disclose is therefore an essential step in the enhanced adherence process for a failing child.
See pages 125-137 of the Consolidated HIV and AIDS Job Aide for full details on the process of disclosure with children.
Ensuring follow up through enhanced adherence and repeat VL testing is imperative. If the second VL result remains more than a 1000 copies/ml, the child’s case should be discussed with your nurse mentor or doctor as soon as possible. If on a PI based regimen non-adherence is more likely than resistance, however discuss with an experienced clinician to decide whether genotyping may be feasible.
PI drugs have a very high genetic barrier and so the virus would need to develop a lot of mutations to be resistant. Therefore, for clients with a high VL, it’s most likely that the client isn’t adhering to treatment rather than that he or she has developed drug resistance.
Treatment Failure Defined for Children
As we discussed last session, treatment failure can be either virologic, immunologic, clinical, or a combination of the three. Last week we focused on how treatment failure is defined for adults. Now, let’s look at how the WHO defines it for children. Tap on each type of failure to read more.
Virologic failure
Viral load greater than 1000 copies/mL based on two consecutive viral load measurements after three months of enhanced adherence counselling.
Immunologic failure
If the child is younger than five years, persistent CD4 count below 200 cells/mm3 or CD4 percentage less than 25. If the child is older than five years, persistent CD4 < 100 cell/mm3 after six months of effective treatment.
Clinical failure
New or recurrent clinical event indicating severe immunodeficiency (WHO stage 3 or 4 clinical condition with the exception of TB) after six months of effective treatment.
When working with children, it’s important to remember that a child’s immune system is still immature and therefore a child will deteriorate faster than an adult. For this reason, we should be even more vigilant when working with children and their caregivers. Low blood levels can also result if a pediatric client gains weight and the dosage isn't adjusted for this gain. Clinicians should be alert if a child has a new stage 2 or 3 OI, recurrent episodes of diarrhoea, respiratory tract infections, new rashes, or shows signs of weight loss. A child who has recurrent illnesses should also trigger a request for a viral load test.
-
Case Study: Mary and Susan (15 min)
You have been the primary nurse/provider for two sisters, Mary and Susan. Mary is 15 years old and has been caring for Susan who is 8 years old. They lost their parents about a year ago. Their mother died of AIDS and their father, who was a heavy drinker, died of liver problems. Mary suspects that her father was also infected with HIV.
Susan was diagnosed HIV positive in 2016 during the initiation phase of TB treatment. She began ABC/3TC/EFV on 02/April/2016 and continued TB treatment. She had difficulty taking all of the pills everyday, and there were times that she missed her follow-up appointments. You suspected she probably missed some of the doses of both treatments. Despite the challenges, Susan completed her TB treatment and her follow-up sputum confirmed she was cured.
At today’s visit, Susan needs a VL test. You know that Susan doesn’t like needles and in the past it has been difficult to palpate her veins. You tell Susan your plan and immediately Mary asks you why her sister needs more blood taken. She tells you that Susan’s viral load test was good the last time it was taken and doesn’t understand why it needs to be done again today. Mary is visibly upset, which makes Susan cry. Susan tells you she doesn’t need the test. “I take all of my medicine now like you told me to do. I haven’t missed any pills, I promise!” she wails. You review her medication record and note that over the past few months, Mary has brought back Susan’s pill bottles more consistently than when she first started on ART. The bottles have been empty for the past three months and before that she had only one or two pills left. Her adherence has been documented in her record as “good” at several visits and 85% and 90% for the first two months after starting ART. You also note that her viral load result six months after starting ART was 868 copies/mL.
Mary comforts Susan, telling her that after she has her blood taken, they will stop at the Tuck shop for a packet of maputi as a reward.
The clinic has been experiencing stock-out of blood collection supplies recently. The facility manager is aware and has notified the district of the items needed. You were told at a recent clinic staff meeting that the supplies were delivered and that all staff would be responsible for monitoring the supplies and notifying the manager when re-ordering was needed. You assemble the blood collection materials needed for Susan’s viral load test.
Susan is sent home with one-month supply of her current ART medication and told to return in a month. You instruct her to continue to take her medication every day and not miss any doses.
As Susan and Mary are leaving, you hear one of your colleagues yelling for help. You immediately assist her with a patient that has collapsed outside your examination room. Once the patient has been transferred to the ED, you’re finally able to take your lunch break before starting the afternoon clinic. Upon returning to the examination room you met Mary and Susan in, you notice that Susan’s blood is still there. You finish completing the laboratory paperwork and send it with the courier who has just arrived for the afternoon pick up.
When Susan’ results come back, you see the following note on the VL results form:
Sample rejected due to clotted specimen and insufficient volume.
Since Susan’s VL sample was rejected, she must get another test at the following month’s visit. You are on leave but you tell your colleague about Susan’s case. Her repeat VL test was done successfully.
Today you are back from leave to see the results:
Viral load: 1005 copies/mL.You review her record and note that your colleague gave a prescription for three months of ART, since Mary told her that it was difficult bringing her sister to clinic every month and Susan’s teacher was upset at how much school she’s missing.
The CHW assigned to Susan has been instrumental in ensuring that she returned to clinic for her monthly appointments and, at the request and permission of Mary, met with Susan’s teacher to go over her clinic schedule. The CHW followed up with the adherence counseling provided by the clinic staff and Susan’s reported no missed doses over the three months of EAC.
Susan returned to clinic today excited to have her viral load test repeated. Mary notices that the tube on the desk is a different color than the last time Susan got tested. You explain to them that the viral load test will be done by a different method called DBS, but the results will still show whether Susan’s medication is working. Mary asks why the clinic is using this test.
Susan’s viral load result was TND copies/mL! You call Mary to let Susan know.
Susan’s case was discussed at the last team meeting in the context of the importance of continued adherence counseling and support. Another nurse shared a concern she had about telling patients that a result was “TND”.
-
Reading: Summary (5 min)
This concludes the final session. In this session, we looked at special populations, focusing on HIV-positive pregnant and breastfeeding women as well as HIV-positive children. We discussed the importance of preventing mother-to-child transmission and looked at the WHO eMTCT validation targets and indicators. We then went over the pregnant and breastfeeding women viral load algorithm and how to manage those who are on versus not on ART. Finally, we concluded the session by delving into the topic of managing HIV-positive children with high viral load.
-
Quiz: Posttest (10 min)
