Welcome to the Parsek Lab
We are a bunch of scientists interested in social aspects of microbial behavior. We use cutting-edge multidisciplinary techniques to address key questions surrounding the molecular mechanisms behind these behaviors. Please contact us and let us know if you are interested in discussing science or collaborating. Enjoy the web site!

Our lab is currently focused on these areas:
The Biofilm Matrix
Biofilm formation has been linked to many chronic bacterial infections. Thus, significant research has been directed towards understanding the basic biology behind biofilm formation. Biofilms produce an extracellular matrix that functions, in part, to hold the community together. Pseudomonas aeruginosa represents a paradigm species for the study of biofilms in the laboratory. The regulation of matrix production and the carbohydrate component of the matrix have both been examined. However, the protein component of the biofilm matrix has been relatively understudied. Our groups identified a biofilm matrix protein for P. aeruginosa, CdrA. CdrA provides structural integrity through extracellular interactions with the matrix polysaccharide Psl. We have additionally identified a novel matrix integrity protein, a Psl-binding lectin, LecB. Outside of matrix proteins that provide structural functions, we predict that matrix associated proteins can play both nutritional and protective roles for the community. Our lab is focused on identifying specific biofilm matrix proteins of P. aeruginosa and the function they play for the biofilm community. We are also interested in the impact of host protein matrix interactions and their ability to impact biofilm survival and pathogenicity.
Biofilms in the Host Environment
A critical element of Pseudomonas aeruginosa pathogenesis is its ability to form biofilms in the lungs of CF patients. Biofilm bacteria produce one or more extracellular polymeric substances (EPS) that act as a scaffold, holding biofilm cells together and to a surface. For some time, alginate has been considered the major polysaccharide of the biofilm EPS matrix. Our studies have indicated that although alginate is a key biofilm matrix component of mucoid strains, it’s not a significant component of the matrix of nonmucoid P. aeruginosa strains, which are the first to colonize CF patients. We discovered that P. aeruginosa has the capacity to encode at least two alternative exopolysaccharides, designated Psl and Pel, which play a critical role in biofilm formation. Psl has been reported to consist of a neutral polymer of a pentasaccharide subunit. Recent discoveries by our group have determined that Pel is a positively charged amino sugar polymer.
Most of the research to date on the biofilm EPS matrix has been conducted in vitro. We know quite a bit regarding the regulation of matrix components, their localization within biofilms, and how they can impact antimicrobial tolerance and resistance to host defenses. However, we know very little regarding matrix composition and assembly in the host. We are at a juncture where we are poised to evaluate and extend our in vitro findings in the context of disease. Towards this end, we will probe in vivo aggregates of P. aeruginosa for evidence of matrix production and localization. Additionally, we will explore the potential of host proteins found in the airways to specifically bind matrix polysaccharides. Our research will provide the foundation for future funding geared towards bridging in vitro biofilm work and understanding the nature of bacterial aggregates observed in vivo.
Surface Sensing by P. aeruginosa
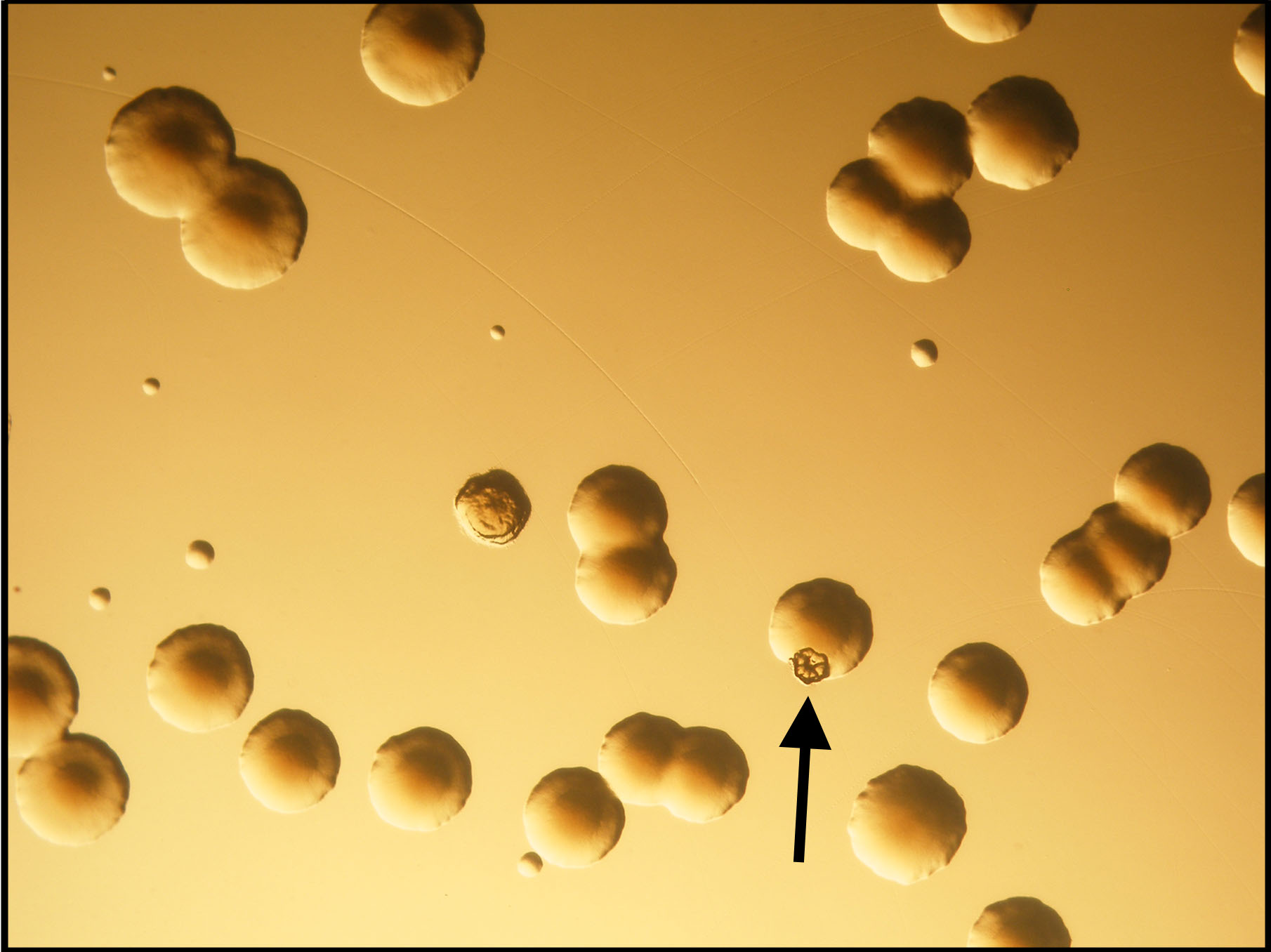
The second messenger signaling molecule cyclic diguanylate monophosphate (c-di-GMP) drives the transition from planktonic to the biofilm mode of growth in a variety of bacteria. Pseudomonas aeruginosa has at least two surface sensing chemosensory systems that produce c-di-GMP in response to growth on a surface. P. aeruginosa biofilm cells have elevated c-di-GMP levels relative to their planktonic counterparts, and mutations in genes involved c-di-GMP synthesis lead to defects in biofilm formation. While factors contributing to the formation of mature biofilms have been well-characterized, early biofilm formation, when a bacterium first senses a surface and transitions from a planktonic state to a surface-attached state, remains largely understudied. Here, we describe how heterogeneity in cellular levels of c-di-GMP among a genetically homogenous population of P. aeruginosa leads to a diversification of bacterial behaviors during surface sensing. Surface attached P. aeruginosa cells specialize into physiologically distinct subpopulations of high c-di-GMP early microcolony “founder” polysaccharide producers and low c-di-GMP early surface explorers, each of which contribute to downstream biofilm formation. Diversification in c-di-GMP levels correlates with clustering of the diguanylate cyclase WspR, and mutations in the Wsp system can disrupt the ability of P. aeruginosa to specialize into founders and explorers. Together, these results suggest that a genetically homogenous population of P. aeruginosa cells engages in a division of labor during early biofilm formation, prior to cells becoming irreversibly attached and forming mature biofilm structures.
Parsek Lab Research
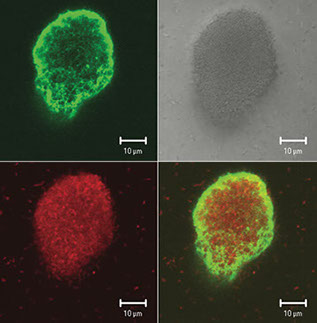
A Pseudomonas aeruginosa aggregate that is stained for all cells (red) and the exopolysaccharide Psl (green). Psl forms a shell around the developing aggregate.
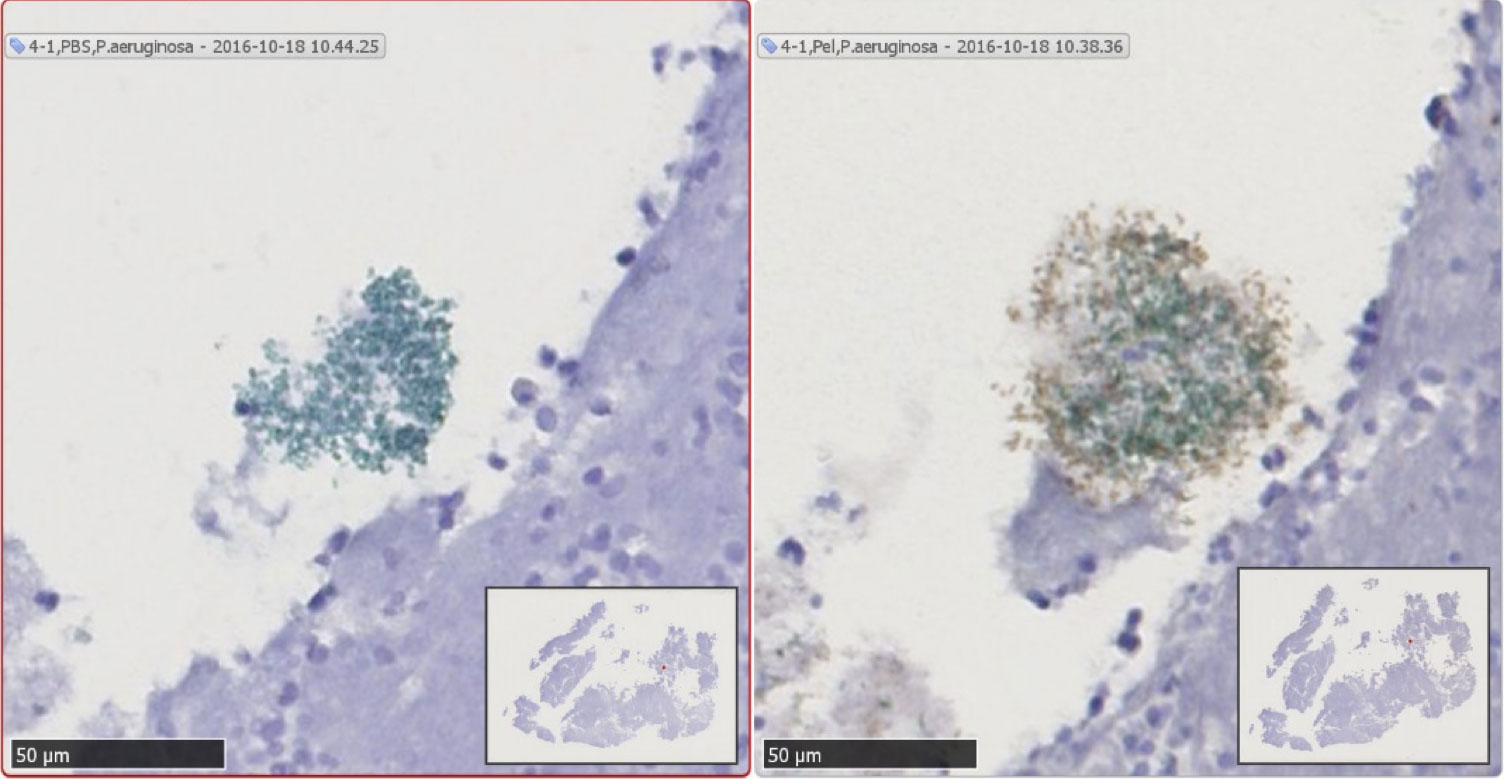
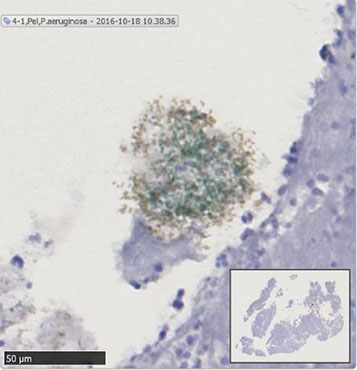
Immunohistochemistry detects P. aeruginosa (green) and Pel (brown) in CF sputum. Slides were imaged with 20X objective and counterstained with hematoxylin (purple) to view host airway material. This figure shows an aggregate of P. aeruginosa stained with P. aeruginosa antisera approximately 50 µm in diameter (green color). This slice was also stained with Pel antisera (brown color). The black bar in the lower left of the panel represents 50 µm.
Surface attached cells displaying elevated levels of GFP (cyan color) and clustering of the Wsp system (yellow, punctate fluorescence). This figure shows that induction of c-di-GMP production in response to a surface corresponds to clustering of the Wsp system.
Matt Parsek, Ph.D., Prinicipal Investigator
parsem@uw.edu
(206) 221-7151
Dr. Parsek received his B.S. in biology from the University of Illinois at Champaign-Urbana. He received his Ph.D. from the lab of Ananda Chakrabarty at the University of Illinois at Chicago Medical Center. He did a post doctoral fellowship at the University of Iowa with Peter Greenberg.
Dr. Parsek has been a Professor in the Department of Microbiology at the University of Washington since 2011. He is a member of the American Academy of Microbiology and was named a Kavli fellow by the National Academy of Sciences.
Catherine Armbruster, Ph.D., Senior Fellow
c4t@uw.edu
(206) 543-3147
B.S. Biology, Mary Baldwin College
M.P.H. Emory University Rollins School of Public Health
Ph.D. Microbiology, University of Washington
As a graduate student co-mentored by Matt Parsek and Luke Hoffman, I investigated how two commonly co-infecting bacterial pathogens, P. aeruginosa and Staphylococcus aureus, interact through their secreted products, altering biofilm formation and pathogenesis. I also examined the emergence of phenotypic heterogeneity within a genetically homogeneous population of P. aeruginosa cells during initiation of biofilm formation, leading to a division of labor in the behaviors associated with early biofilm formation: surface exploration and polysaccharide production. Together, these studies provide a glimpse into P. aeruginosa sociality and contribute to the growing body of literature detailing how bacterial social interactions drive the composition and function of biofilms.
Julia Dreifus
jdreifus@uw.edu
(206)543-3147
B.S. Biotechnology, Rutgers University
My research interests revolve around microbial biofilms, specifically the role that biofilm matrix components play in disease. I work on evaluating the roles of Pseudomonas aeruginosa biofilm matrix components in chronic cystic fibrosis infections. The Pseudomonas biofilm matrix contains three exopolysaccharides and I am working to uncover interactions between these products and host components, and to evaluate the effects of these interactions on antimicrobial resistance. I am also interested in investigating interactions involving the Staphylococcus aureus exopolysaccharide PIA. In addition to my lab duties, I am responsible for providing a steady supply of baked goods.
Holly Jacobs, B.A.
hmjacobs@uw.edu
B.A., Biochemistry & Molecular Biology, The College of Wooster
I’m interested in the extracellular matrix (ECM) proteome of Pseudomonas aeruginosa. I am currently investigating what aspects of the ECM proteome can be attributed to the three distinct exopolysaccharides (Pel, Psl, and alginate) which are produced in P. aeruginosa biofilms.
Michael Matwichuk
B.S Microbiology (Winter 2018)
mike2017@uw.edu
(206)543-3147
I will be working on proteins that stabilize the biofilm matrix. One of these proteins is the self-produced lectin, LecB. LecB stablilizes P. aeruginosa biofilms through binding of the exopolysaccharide Psl. In addition, I will be analyzing the role of cheaters in P. aeruginosa biofilm formation.
Jessica Parker-Gilham
jesspgik73@gmail.com
B.S. Microbiology, University of Washington
My passion is bacterial communities called biofilms and how they form and working in the Parsek lab has taught me the tools I need to pursue this passion. My main project is studying Pseudomonas aeruginosa c-di-GMP dynamics in the very early stages of biofilm formation. To do this I work with flow cells and the confocal microscope a lot!
Daniel Passos da Silva, Ph.D., Senior Fellow
passosds@uw.edu
(206)543-3147
B.Sc in Microbiology and Imunnology. Federal University of Rio de Janeiro, UFRJ, Rio de Janeiro, Brazil
M.Sc. in Biochemistry. Federal University of Rio de Janeiro, UFRJ, Rio de Janeiro, Brazil
Ph.D. in Genetic Engineering and Biotechnology. International Centre for Genetic Engineering and Biotechnology, ICGEB, Trieste, Italy.
I am a microbiologist fascinated by bacterial communities. Currently I am studying the role of lectins for Pseudomonas aeruginosa biofilm formation.
Courtney Reichhardt, Ph.D., Senior Fellow
crreich@uw.edu
(206) 543-3147
B.S., Chemistry, Montana State University
Ph.D., Chemistry, Stanford University (Advisor: Professor Lynette Cegelski)
My postdoctoral research uses both biophysical and microbiological techniques to understand how the structure and function of individual biofilm matrix components contribute to the changes that biofilms undergo throughout their lifecycle. Specifically, I am investigating the maintenance and regulation of Pseudomonas aeruginosa biofilm structural integrity with a specific focus on the role of the biofilm matrix protein called CdrA.
Delaney Townsend
dtown1@uw.edu
(206) 543-3147
B.S. Microbiology, University of Washington
B.A. Biochemistry, University of Washington
I recently graduated from the University of Washington with a BS in Microbiology and MCD Biology and an BA in Biochemistry. Earlier, I was working on a project looking at the expression levels of LecB during biofilm formation using a GFP reporter. Now I am beginning a new project studying the function of two proteins PA2440, a putative deacetylase, and PA2441, a hypothetical protein, and how they influence biofilm phenotypes.
Cynthis Wong
cynthis.wong@gmail.com
(206) 543-3147
B.S., Microbiology
I am working closely with Courtney to explore the role of the matrix protein CdrA in Pseudomonas aeruginosa biofilms. Specifically, I am investigating the functional impact of interactions between CdrA and matrix polysaccharides.
Parsek People










News from the Parsek Lab…
Here’s the latest:
Parsek/Woz retreat celebrates its 10th year!
The Parsek and Wozniak labs celebrated their 10th annual summer retreat August 20-23, 2017. We were joined by the Howell, Harrison, and Tseng labs. Our guest speaker was Valerie Waters from U Toronto — Thanks, Valerie!! The “golden pipette” competition was won by the Parsek Lab. Check out the cool group photo that was taken during the eclipse.



Congratulations Daniel and Catherine!
Both were selected to give oral presentations at the recent Eurobiofilms conference in Amsterdam. According to the twitter feeds, they did a great job. Daniel talked about the role of lectins in P. aeruginosa biofilm formation, while Catherine presented on heterogeneity in surface sensing. Cheers Daniel and Catherine!
Our Collaborators
Lynne Howell, University of Toronto
Joe Harrison, University of Calgary
Other Links
Links
Our lab finds these links useful: