|
| Cardiac Signaling |
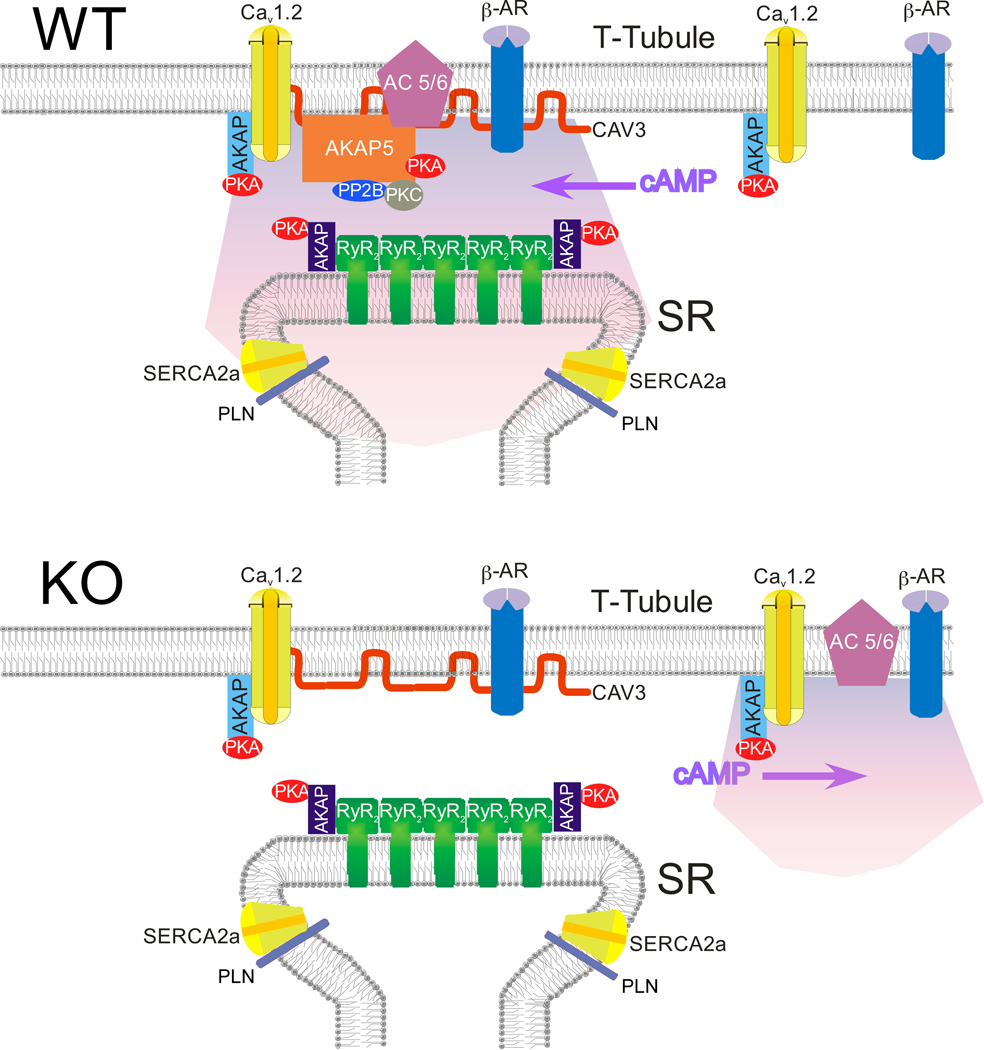
Sympathetic stimulation of the heart elevates cAMP levels leadingto the rapid phosphorylation of several protein targets by proteinkinase A (PKA). Immediate consequences of this phosphorylation include increased Ca2+ entry through voltage-gated Ca2+ channels(CaV1.2), Ca2+ release from sarcoplasmic reticulum (SR) ryanodinereceptors, and Ca2+ reuptake into the SR by the calcium ATPase(SERCA). Enhanced Ca2+ cycling amplifies the force ofcontraction and rate of relaxation, substantially improving cardiacperformance. Timely and specific phosphorylation of PKAsubstrates is made possible by scaffolding molecules called AKinase Anchoring Proteins (AKAPs) which bind directly to PKA, phosphatases, ion channels, and other proteins and membranes tocoordinate discrete molecular signaling complexes. SeveralAKAPs are expressed in the heart. Disruption of all AKAP-PKAbinding abolishes the effect of sympathetic stimulation on Ca2+cycling. Our work utilizes AKAP knockout/mutant mice to identify specific AKAP(s) that are essential for CaV1.2 andPLB/SERCA regulation. Genetically targeted AKAPs includeAKAP7 (15/18), AKAP5 (79/150), and AKAP1. Model of AKAP5 function in Cardiomyocytes from Nichols et al Circulation Research 2010 |
Contraction An isolated cardiomyocyte is loaded with Furo-4 (a calcium dye), and pulsed at 25V, 1Hz. This is a measurement of the calcium transient induced at this frequency. |
|
Sparks An isolated cardiomyocyte is loaded with Furo-4 (a calcium dye) and recorded without stimulation. This video looks at Calcium movement and the spontaneous function of the Ryanidine Receptors. |