Introduction:
After doing the Core lab or the Development lab you will have reached the pluteus stage of sea urchin development. To carry the baby sea urchins further you need to start feeding them. If all goes well, you will be ready for metamorphosis in about 30-40 days.
See Pluteus to Urchin animation and Embryo Culture.
Simple method:
OK, this is the easy way, but it does not work as well and you probably will not make it all the way to metamorphosis. You will, however, be able to extend the pluteus stage out for several weeks. This is more than enough time to do additional experiments and observations.
You will need to order some Isocrysis micro algae paste. We have some students working with the various algal pastes and the best one to date seems to be the "Tahitian Blend" from Brine Shrimp Direct (see Suppliers).
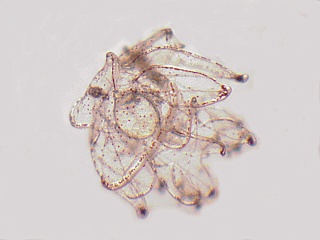
This is an excellent food for lots of filter feeders in your aquarium, from mussels, clams, tunicates, etc. and a single vial goes a long way.
Isocrysis is a golden brown flagellate of approximately 5-6 microns is size. There are two other species listed, Tetraselmis (8-16 micron) and Nannochloropsis (2-4 micron). They do best with Isocrysis, but because the Tetraselmis is bigger it shows up in the gut better for observations on feeding behavior. The Nannochloropsis is too small to see well and the plutei do not do well on it either.
To use:
Grow a 0.1% culture of sea urchin larvae for 3-5 days post fertilization. At this point they will be ready to feed. The Isocrysis paste has 8.6 billion cells/ml and we need to dilute this to 3000 cells/ml. One drop of paste contains approximately 430 million cells. If we add one drop of paste to one liter of seawater we now have 430,000 cells / ml. One drop of this diluted stock will do 7 ml of culture. I realize that you will be throwing a lot of the algae out, but they are not very expensive to begin with. You may be able to freeze the intermediate stock. We have not tried this. As with all feeds, it is important not to over-feed as excess food will rot and increase bacterial problems. Bacteria buildup is the #1 reason a culture fails. By the time they are ready to metamorphose, you should be down to a maximum of about 100 per liter. Better to underfeed a bit than to over feed. #2 is over crowding. You will have to "thin" the cultures out as the plutei get bigger.
At least once per week, and possibly more often, you will need to change the seawater in the cultures. (more often if you have a bacterial buildup). The best way to do this is with a very slow siphon with a 110um mesh on the end of a 50 ml plastic conical centrifuge tube. You may also want to siphon the dead larvae off the bottom of the culture. See Embryo Culture.
Better, but harder method:
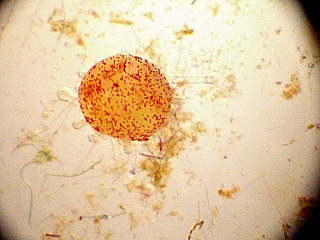
Here we use live food. This means culturing the food, a Rhodamonas algae (here we want Rhodomonas salina – see Suppliers).
The media is prepared as follows (adapted from the Provasoli web site, see References):
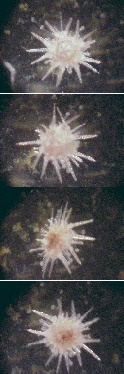
1 week old juvenile
executing a turnover
f/2-Si Medium
To 950 mL filtered seawater add:| Quantity | Compound | Stock Solution |
|---|---|---|
| 1.0 mL | NaNO3 | 75.0 g/L dH2O |
| 1.0 mL | NaH2PO4.H2O | 5.0 g/L dH2O |
| 1.0 mL | f/2 Trace Metal Solution | (see recipe below) |
| 0.5 mL | f/2 Vitamin Solution | (see recipe below) |
Make final volume up to 1.0 L with filtered seawater. Autoclave after all additions.
f/2 Trace Metal Solution
To 950 mL distilled H2O add:| Quantity | Compound | Stock Solution |
|---|---|---|
| 3.15 g | FeCl3.6H2O | - |
| 4.36 g | Na2EDTA.2H2O | - |
| 1.0 mL | CuSO4.5H2O | 9.8 g/L dH2O |
| 1.0 mL | Na2MoO4.2H2O | 6.3 g/L dH2O |
| 1.0 mL | ZnSO4.7H2O | 22.0 g/L dH2O |
| 1.0 mL | CoCl2.6H2O | 10.0 g/L dH2O |
| 1.0 mL | MnCl2.4H2O | 180.0 g/L dH2O |
Make final volume up to 1.0 L with dH2O. Autoclave.
f/2 Vitamin Solution
To 950 mL dH2O add:| Quantity | Compound | Stock Solution |
|---|---|---|
| 1.0 mL | Vitamin B12 (Cyanocobalamin) | 1.0 g/L dH2O |
| 10.0 mL | Biotin | 0.1 g/L dH2O |
| 200.0 mg | Thiamine HCl | - |
(You may be able to get small quantities of these vitamins at a health food store) Make final volume up to 1.0 L with dH2O. Filter sterilize into plastic vials and store in refrigerator.
Note: vitamin B12 and Biotin are obtained in a crystalline form. When preparing the Vitamin B12 Stock Solution allow for approximately 11% water of crystallization (For each 1.0 mg of Vitamin B12 add 0.89 mL dH2O). When preparing the Biotin Stock Solution allow for approximately 4% water of crystallization (for each 1.0 mg of Biotin add 9.6 mL dH2O) .
You need to use sterile technique thoughout. The cultures are allowed to sit at room temperature AWAY from open windows (they don't like direct sunlight). Fluorescent room lights are enough.
A hemacytometer can be used to assess culture denisty. You want to feed the plutei at a final algae concentration of 3000 cells/ml. Since the media is toxic to the sea urchin larvae, the algae need to be centrifuged gently (200g for 5 minutes) and the f2-Si media removed. The cells are then resuspended in filtered seawater and fed with the necessary dilution to the larvae.
Maintaining sterile algae cultures is not trivial and this should not be attempted without experience and patience.
Metamorphosis:
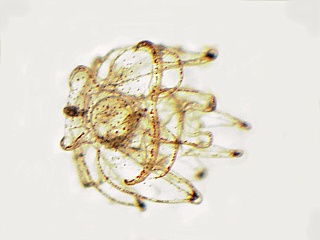
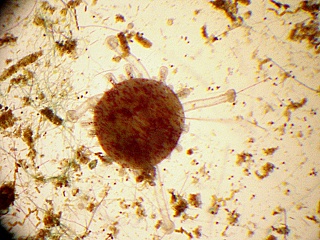
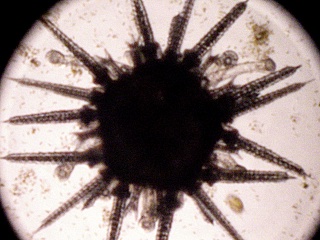
In order for sea urchins to metamorphose, they need a suitable substrate to settle on. They like surfaces that have a bacterial slime covering them. This is accomplished by letting small petri dishes or slides sit in an established aquarium or running seawater for several days to several weeks (lots of slime and diatoms). Plutei at the right stage (10 arm stage) will metamorphose in these dishes in 1-3 hours, turning into minature sea urchins with 5 tube feet and five sets of two spines.
Beyond:
At this point the baby sea urchins need a diatom, Nitzschia sp., to grow further. This is probably beyond what can be accomplished in a high school or junior college setting (for more details, see Hinegardner and Tuzzi).

References:
- Guillard R.R.L. and Ryther J.H., "Studies of marine planktonic diatoms. I. Cyclotella nana Hustedt and Detonula confervacea Cleve.", Canadian Journal of Microbiology, 8: 229-239, 1962.
- Guillard R.R.L., "Culture of phytoplankton for feeding marine invertebrates", Culture of Marine Invertebrate Animals, eds: Smith W.L. and Chanley M.H. (New York: Plenum Press, 1975), pp 26-60.
- Ralph Hinegardner and Mary Tuzzi, "Laboratory Culture of the Sea Urchin Lytechinus pictus", from the book Laboratory Animal Management, Marine Invertebrates, National Academy Press, 1981.
The work on this page, including help in taking the images, is the result of the efforts of Stacey Woo, an undergraduate in our lab doing an honors thesis.
