Research Highlight #10:
A Genetic Approach for Controlling Binding and Orientation of Proteins on Nanoparticles
A. Sengupta, C.K. Thai, M.S.R. Sastry, J. Matthaei, D.T. Schwartz, E.J. Davis & F. Baneyx;
Chemical Engineering, University of Washington
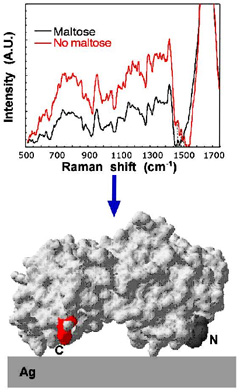
Although silver nanoparticles are excellent surface enhancers for Raman spectroscopy, their use to probe the conformation of large proteins at interfaces has been complicated by the fact that many polypeptides adsorb weakly or with a random orientation to colloidal silver. To address these limitations, we sought to increase binding affinity and control protein orientation by fusing a silver-binding dodecapeptide termed Ag4 to the C-terminus of maltose-binding protein (MBP), a well-characterized model protein with little intrinsic silver binding affinity. Quartz crystal microbalance measurements conducted with the resulting MBP-Ag4 fusion protein revealed that its affinity for silver was at least one order of magnitude higher than a control protein, MBP2, containing a non-silver specific C-terminal extension. Under our experimental conditions, MBP-Ag4 surface-enhanced Raman (SERS) spectra exhibited 2-4 fold higher signal-to-background relative to MPB2 and contained a number of amino acid-assigned vibrational modes that were either weak or absent in control experiments performed with MBP2. Changes in amino acid-assigned peaks before and after MBP-Ag4 bound maltose were used to assess protein orientation on the surface of silver nanoparticles. This genetic approach may prove useful to study the orientation of other proteins on a variety of SERS-active surfaces, to improve biosensors performance and to control functional nanobiomaterials assembly.
Contact F. Baneyx, for more details, baneyx@u.washington.edu.
Langmuir, 24 2000-2008 (2008).



