Research Highlight #18:
Effect of Molecular Conformations on the Adsorption Behavior of Solid-Binding Peptides
M. Hnilova, E.E. Oren, U.O.S. Seker, B.R. Wilson, S. Collino, J. S. Evans C. Tamerler, and M. Sarikaya;
Materials Science & Engineering and Microbiology, University of Washington
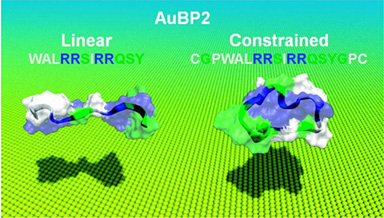
Despite extensive recent reports on combinatorially selected inorganic-binding peptides and their bionanotechnological utility as synthesizers and molecular linkers, there is still only limited knowledge about the molecular mechanisms of peptide binding to solid surfaces. Here we discuss the effects of constraints imposed on FliTrx-selected gold-binding peptide molecular structures upon their quantitative gold-binding affinity. The two gold-binding peptides (AuBP1 and AuBP2) exhibit a range of binding affinities to and adsorption kinetics on gold surfaces, with the equilibrium constant, Keq, varying from 2.5 × 106 to 13.5 × 106 M-1. Both circular dichroism and molecular mechanics/energy minimization studies reveal that each of the four peptides has various degrees of random coil and polyproline type II molecular conformations in solution. We found that AuBP1 retained its molecular conformation in both the c- (constraint) and l- (linear) forms, and this is reflected in having similar adsorption behavior. On the other hand, the c- and l-forms of AuBP2 have different molecular structures, leading to differences in their gold-binding affinities.
Contact C. Tamerler, for more details, candan@u.washington.edu.
Langmuir, 24 12440-12445 (2008).



