Research Highlight #21:
Chemically Controlled Self-Assembly of Enzyme Nanorings
T. F. Chou, C. So, B. R. White, J. C. T. Carlson, M. Sarikaya and C. R. Wagner;
Materials Science & Engineering and Microbiology, University of Washington
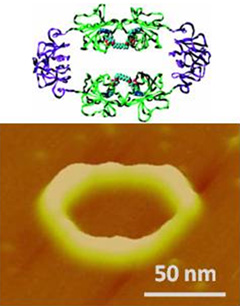
We have discovered a method for the preparation of stable and homogeneous protein polygons, or nanorings. In particular, dihydrofolate reductase (DHFR) molecules when fused together by a peptide chain of variable length were found to spontaneously self-assemble in to protein macrocycles after treatment with a dimeric enzyme inhibitor (MTX), that efficiently dimerizes DHFR. The size of the nanoring (8-30 nm diameter) was found to be dependent on the length of the linker peptide between the two DHFRs. To expand the potential size of protein nanoring self-assembly, we hypothesized that replacement of a portion of the amino acid linker by a rigid protein might favor formation of larger nanostructures. The protein nanorings could potentially be used as molecular substrates for biosensing, multi-functional probing, and electromagnetic nanoresonators.
Contact C. R. Wagner, for more details, wagne003@umn.edu.
ACS Nano, 2 2519-2525 (2008).



