Research Highlight #3:
Nonequilibrium Synthesis and Assembly of Hybrid Inorganic-Protein Nanostructures Using an Engineered DNA Binding Protein
Haixia Dai, Woo-Seok Choe, Corrine K. Thai, Mehmet Sarikaya, Beth A. Traxler, François Baneyx, and Daniel T. Schwartz;
Chemical Engineering, Materials Science & Engineering, and Microbiology, University of Washington
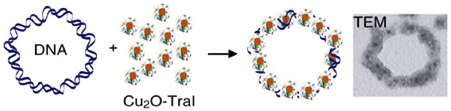
We show that a protein with no intrinsic inorganic synthesis activity can be endowed with the ability to control the formation of inorganic nanostructures under thermodynamically unfavorable (nonequilibrium) conditions, reproducing a key feature of biological hard-tissue growth and assembly. The nonequilibrium synthesis of Cu2O nanoparticles is accomplished using an engineered derivative of the DNA-binding protein TraI in a room temperature precursor electrolyte. The functional TraI derivative (TraIi1753::CN225) is engineered to possess a cysteine-constrained 12-residue Cu2O binding sequence, designated CN225, that is inserted into a permissive site in TraI. When TraIi1753::CN225 is included in the precursor electrolyte, stable Cu2O nanoparticles form, even though the concentrations of [Cu,+] and [OH–] are at 5% of the solubility product (Ksp,Cu2O). Negative control experiments verify that Cu2O formation is controlled by inclusion of the CN225 binding sequence. Transmission electron microscopy and electron diffraction reveals a core–shell structure for the nonequilibrium nanoparticles: a 2 nm Cu2O core is surrounded by an adsorbed protein shell. Quantitative protein adsorption studies show that the unexpected stability of Cu2O is imparted by the nanomolar surface binding affinity of TraIi1753::CN225 for Cu2O (Kd=1.2x10–8 M), which provides favorable interfacial energetics (–45 kJ/mol) for the core-shell configuration. The protein shell retains the DNA-binding traits of TraI, as evidenced by the spontaneous organization of nanoparticles onto circular double stranded DNA.
Contact D. Schwartz for more details, dts@u.washington.edu
Journal of the American Chemical Society, 127(44), 15637-15643, 2005.



