Research Highlight #6:
Conformational Control of Designer Protein-Inorganic Adhesion
W.S. Choe, M.S.R. Sastry, C.K. Thai, D.T. Schwartz, & F. Baneyx;
Chemical Engineering, University of Washington
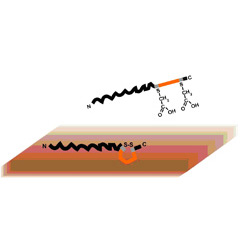
Combinatorial selection of peptides that bind technological materials has emerged as a valuable tool for room-temperature nucleation and assembly of complex nanostructured materials. However, the parameters that control peptide-solid binding are poorly understood. Previously, we reported a statistically significant enrichment in paired arginines (RR) among a family of disulfide-constrained cuprous oxide binding peptides, and hypothesized that this is a key motif for metal oxide binding. Using site-directed mutagenesis of a DNA binding protein engineered with one such cuprous oxide-binding loop, we found that the RR motif only played a minor role in adhesion. On the other hand, presentation of the peptide in a disulfide-constrained loop (the conformation present during combinatorial selection) was crucial for metal oxide binding. These results suggest that caution should be exerted when extrapolating from statistical data and that, in some cases, conformation is more important than composition in determining peptide-inorganic adhesion. From a practical standpoint, an ability to completely abolish binding of designer proteins to their cognate materials by reducing the disulfide bond opens the door to the design of reconfigurable hybrid materials in which solid binding activity can be addressed (electro)chemically or enzymatically.
Contact F. Baneyx, for more details, baneyx@u.washington.edu.
Langmuir, 23 11347-11350 (2007).



