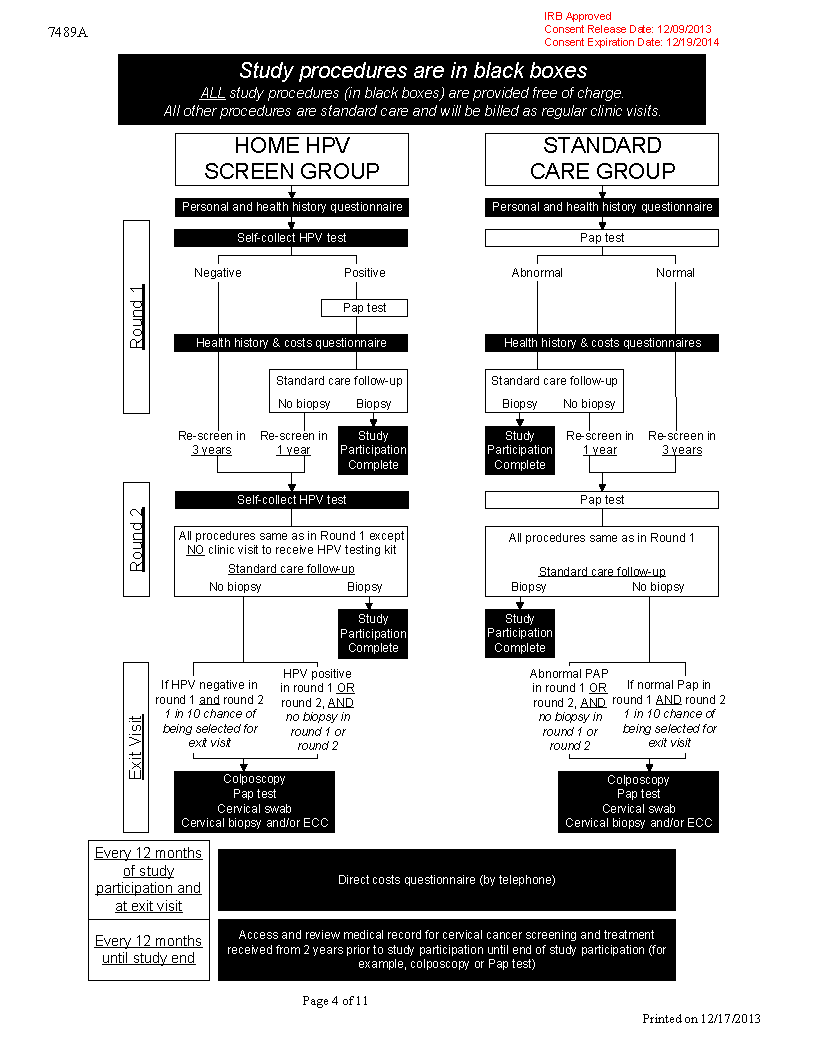
You will be assigned to a study group.
There are two groups of patients in this study. We will give different treatments to different groups, and compare the results. This is how we hope to find out if self-collected at home HPV testing is effective, cost-effective, and/or acceptable to patients.
A computer program will place you in one of the study groups. You cannot choose the group you will be in. You will have a one-in-two chance of being placed in a given group.
If both the HPV and Pap tests return negative, you will no longer be a participant in the study. You will also have a one-in-ten chance of being invited for an in-person exit visit. A computer program will make the selection. You cannot choose the group you will be in.
What research tests, procedures, and treatments are part of this study?
If you decide to join this study, depending on the group you are placed in, we may ask you to do all or some of these tests and procedures:
Medical records history. We will obtain the following from your medical records: (1) cervical cancer related screening and treatment from 2 years PRIOR to the study until the end of your study participation (2) HPV vaccination history.
Self-collect vaginal specimen for HPV testing. (We will ONLY ask you to perform this, if you are placed in the home HPV screening group) We will ask you to wash your hands and then push a cotton swab into your vagina as far as you can with no pain. Rotate three times and take the swab out. Put the swab in a specimen cup. Repeat the sample collection a the second swab.
Colposcopy. If the results of your screening test (Pap test or self-collected at-home HPV test) are abnormal, the study will offer the standard care follow-up of colposcopy. During the colposcopy, the provider will examine your cervix thoroughly using a colposcope (a big microscope for looking at the cervix).
- Cervical biopsy. During the colposcopy, the provider will do a biopsy of your cervix. If the provider recommends it AND you agree, the provider will numb the area using a small needle to inject medication. A biopsy means taking a small piece(s) of tissue from your cervix and testing it. A biopsy can detect changes that a Pap test may miss. The tissue will be examined for the presence of cancer or changes that indicate that you might develop cancer if you are not treated. You will be counseled about the meaning of the results.
- Endocervical curettage (ECC). During the colposcopy, if the provider recommends it and you agree, the provider may take a sample of your cervix using a spoon-shaped instrument called a curette. The provider will scrape a sample of your cervix with the curette. We will test the sample for abnormal or cancerous cells. You will be counseled about the meaning of the results.
Loop electrosurgical excision (LEEP). If the cervical biopsy shows precancerous changes that require treatment, the study clinician will advise you on the type of treatment that is appropriate. The study will offer Loop Electrosurgical Excision Procedure (LEEP) treatment, a standard procedure in which precancerous areas in the cervix are removed using a fine wire loop with electric current. If you require treatment other than LEEP, the study clinician will refer you to appropriate clinics for care. If you are diagnosed with cervical cancer, the study clinicians will notify you immediately and refer you to a gynecological oncologist for immediate follow-up and treatment.
Questionnaires. Some of the questions may be sensitive. If a question makes you feel uncomfortable, you may choose not to answer.
- Personal and health history questionnaire. We will ask you about yourself and your health history. For example, what is the highest level of schooling you have completed, or have you used contraceptives These questions are part of a standard questionnaire for everyone in the study.
- Health history and costs questionnaire. We will ask you about your health history, your Pap screening history, and any out-of pocket costs related to your clinic visit. For example, have you been sexually active in the last six months, or did you feel any embarrassment when you had your last Pap test, or did you have to take time off work to attend the clinic visit. These questions are part of a standard questionnaire for everyone in the study.
If you are placed in the home HPV screening group, we will also ask you about the ease of using the homebased collection kit, preference for at-home screening compared vs. clinic-based screening, preference for the HPV vs. Pap test, time taken to read, perform and mail the self-collection test as well as any out-of-pocket costs for completing the test.
- Direct costs questionnaire. We will ask you about any additional cervical cancer screening that occurred outside of the University of Washington Medicine system and why these visits occurred.
Exit Visit. (only if invited to Exit Visit; see study diagram on next page)
- Colposcopy. The provider will examine your cervix thoroughly using a colposcope (a big microscope for looking at the cervix).
- Pap test. During the colposcopy, the provider will take a sample of your cervix using a soft brush. We will use this sample to make a Pap test. We will test the sample for abnormal or cancerous cells.
- Cervical swab. During the colposcopy, the provider will take one swab of your cervix to test for HPV and other factors that may increase the risk of cervical cancer. This specimen will be used for research purposes only and you will not receive the results of this testing.
- Cervical biopsy. During the colposcopy, the provider will do a biopsy of your cervix. If the provider recommends it AND you agree, the provider will numb the area using a small needle to inject medication. A biopsy means taking a small piece(s) of tissue from your cervix and testing it. A biopsy can detect changes that a Pap test may miss. The tissue will be examined for the presence of cancer or changes that indicate that you might develop cancer if you are not treated. You will be counseled about the meaning of the results.
- Endocervical curettage (ECC). During the colposcopy, if the provider recommends it and you agree, the provider may take a sample of your cervix using a spoon-shaped instrument called a curette. The provider will scrape a sample of your cervix with the curette. We will test the sample for abnormal or cancerous cells. You will be counseled about the meaning of the results.
Schedule of study events
How long will I be in this study?
We think you will be in this study for about 1 to 4 years. The total time depends on the results of your cervical cancer screening tests. For example, if all of your screening results are normal, study participation will be longer, since it may be up to 3 years between screening visits.
Round 1 will take between 1 day and a few weeks, depending on your screening results and how soon you can schedule any recommended follow-up examinations and/or treatment.
Round 2 will occur 1 to 3 years later, depending on your Round 1 screening results.
The exit visit will occur 3 to 12 months after round 2. The exit visit date will depend on when you complete Round 2..
The study doctor or your doctor may take you out of this study at any time. This would happen if:
- They think it is in your best interest not to continue in the study.
- You are unable or unwilling to follow study procedures.
- The whole study is stopped.
If you are thinking about dropping out of this study, please tell the study doctor.
If you leave the study, your test results and information cannot be removed from the study records.