 The Cell Cycle and DNA Replication
The Cell Cycle and DNA Replication
Recent research in the Laird Lab addresses several important concepts in Cell Biology. 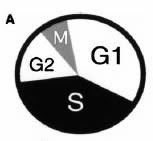
The concept of a "cell cycle" originated in the work of Howard and Pelc (1953), who used early techniques of radiolabeling to determine that most DNA synthesis occurred in a restricted period, now termed "S", for synthesis, phase. Little DNA synthesis occurred immediately following mitosis (G1, for the first gap phase) and just prior to mitosis (G2, for second gap phase). The cell cycle concept became codified in text books (see figure A, from Widrow et al., 1997).
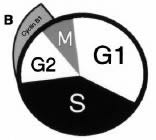
Our finding that DNA replication at the fragile X locus can occur very late in the cell cycle (Hansen et al., 1993), encouraged us to examine further the question of very late DNA replication. R.J. (RJ) Widrow, then a MD-PhD student in the lab, developed a method to separate cells that were at very late stages in the cell cycle by using levels of cyclin B1, which were previously thought to increase only after DNA replication was complete (see figure B, from Widrow et al., 1997).

RJ used his new method to characterize the timing of replication of other late-replicating sequences including several that were known not to be associated with fragile sites. His data led to a new view of the cell cycle, in which the G2 period is much shorter than previously thought. In some cells, a true G2 phase with no DNA replication may not exist (see figure C, from Widrow et al., 1997).
The Laird Lab will be returning to questions of replication timing, aided by their new methods for analysis of DNA methylation.
![]()
The cell biological concept that the timing of DNA replication is locus-specific and functionally important originated with the finding that the inactive-X chromosome in female placental mammals is late-replicating, and corresponds to the hypercondensed "Barr body" (Gartler and Burt, 1964; German, 1962; Taylor, 1960). This finding was extended by Goldman and Holmquist (1984) and others to a broader set of late-replicating, transcriptionally silent chromosome regions. Our work on fragile X syndrome provided an important example for this concept, and demonstrated that replication timing can be altered mutationally and epimutationally (Hansen et al., 1993), with important phenotypical consequences (Laird et al., 1987 and see Human Disease).
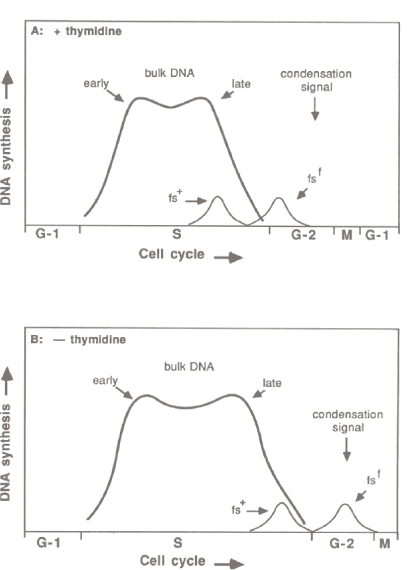
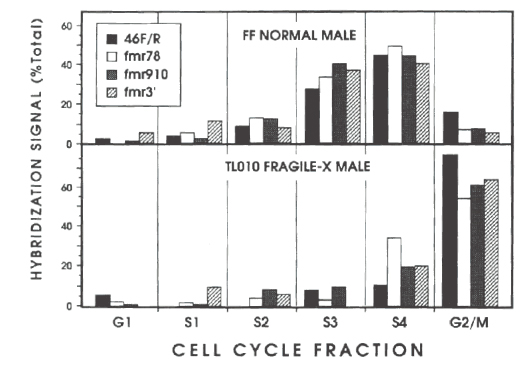
Figure at left, from (Laird et al., 1987): Observed timing of DNA synthesis of fragile site DNA during the cell cycle. Replication of both normal alleles (fs+) and fragile alleles (fsf) are indicated under normal thymidine conditions (A) and under conditions of thymidine deprivation used to induce the fragile site at Xq27.3 (B).
Figure above, from (Laird et al., 1993, Hansen et al., 1993): Timing of replication of FMR1 and flanking sequences in cells from normal (top) and affected (bottom) males.
The Laird Lab has published broadly in others areas of Cell Biology.