What initiates the early phases of neurodegeneration and its progression through brain cells? Research led by Brian Kraemer, PhD, Research Associate Professor in the UW Medicine Division of Gerontology, has made headway on this burning research question, which promises to reveal therapeutic targets.
The Kraemer Lab team focuses on understanding the process by which certain proteins, such as tau and TDP-43, crumple into aggregates. They are the signature brain pathologies in variants of frontotemporal lobar degeneration (FTLD) and amyotrophic lateral sclerosis (ALS). FTLD, commonly called FTD, is the leading cause of dementia in people under 65 years of age (early onset).

Brian Kraemer, PhD
When scientists look at diseased brain tissue under the microscope, they observe that these clumps contain large amounts of “phosphorylated” protein.
Phosphorylation normally directs a protein’s shape alterations, activity, and function in cells. Enzymes called kinases perform this biological modification. Now, the Kraemer Lab has new insights into how the phosphorylation of TDP-43 goes awry in the initial stages of neurodegenerative diseases.
This line of research started when the Kraemer Lab's Nicole Liachko, PhD, Acting Assistant Professor in the Division of Gerontology, made a discovery about kinases known to phosphorylate tau, called tau tubulin kinases 1 and 2 (TTBK1/2). In 2014, she found that these kinases also phosphorylate TDP-43 and seem to regulate the formation of the pathological version.
"We want to find out why the phosphorylation of TDP-43 is going wrong," says Laura Taylor, Graduate Research Associate in the lab. "Ideally, if we could ever prevent that modification, we would prevent these diseases from happening in the first place."

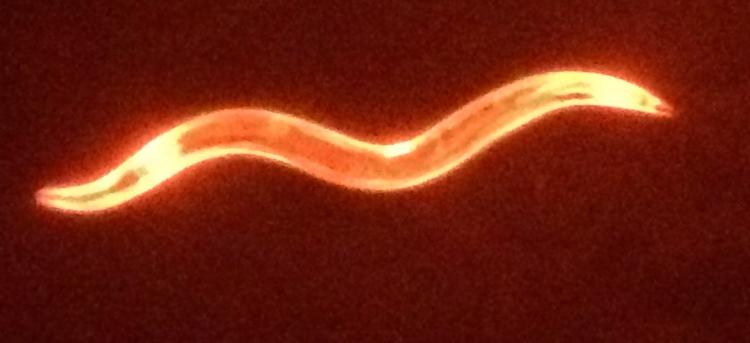
The spinal cord neurons in this C. elegans nematode have been tagged with florescent proteins. Kraemer Lab.
To explore the role of these kinases in FTLD, the team has the help of one of the simplest creatures imaginable. They use the C. elegans worm as a model of TDP-43 proteinopathy. These tiny 1-mm worms serve as a useful model of neurodegeneration because they are easily genetically manipulated to express the human TDP-43 protein. “They have exactly 302 neurons, so it's easy for us to keep track of when any of them begin to grow abnormally or die,” says Taylor.
These worms can't handle the human version of TDP-43, which turns into phosphorylated aggregates in their cells. They lose neurons and develop motor symptoms reminiscent of ALS. And when the researchers overexpress (ramp up) the human kinases TTBK1/2 in this worm line, the phosphorylation of TDP-43 happens sooner and at higher levels. The worms show more neurodegeneration and cell deformities. They also get sicker, faster. They develop obvious motor defects earlier on, and have shorter life spans.

Laura Taylor
Earlier this fall, Taylor traveled to Munich, Germany for the 10th International FTLD Conference to present these findings. In a "data blast," she reported this new evidence that hyperactivation of the TTBK1/2 kinases leads to early and severe disease progression in the worm model. She also noted that her team finds increased levels of TTBK1/2 in postmortem brain tissue of people with FTLD.
Taylor says it was exciting to share this work at the world's most important conference for FTLD researchers because it's some of the first in the field. "It's a novel concept that these kinases are integral to FTLD-TDP-43 disease progression," she says.
Kraemer, who has started to investigate these processes in the more complex mammalian nervous systems of mice, thinks that TTBK1/2 could provide an elegant solution for the task of finding therapies for the pathologically diverse variants of FTLD.
“What I think is really important about TTBK1/2 is that these kinases are hyperactivated in both FTLD-tau cases and FTLD-TDP-43 cases. It’s a common feature in two pathologically distinct diseases," says Kraemer. "That suggests that TTBK1/2 can serve as a therapeutic target, not just for one, but for all forms of FTLD.”
—Genevieve Wanucha
GIF: Movement of C. elegans. Credit: Bob Goldstein, UNC